Effect of Incomplete Cryoablation and Matrix Metalloproteinase Inhibition on Intratumoral CD8+ T-Cell Infiltration in Murine Hepatocellular Carcinoma
- PMID: 38349244
- PMCID: PMC10902598
- DOI: 10.1148/radiol.232365
Effect of Incomplete Cryoablation and Matrix Metalloproteinase Inhibition on Intratumoral CD8+ T-Cell Infiltration in Murine Hepatocellular Carcinoma
Abstract
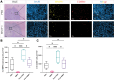
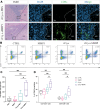
Background Image-guided tumor ablation is the first-line therapy for early-stage hepatocellular carcinoma (HCC), with ongoing investigations into its combination with immunotherapies. Matrix metalloproteinase (MMP) inhibition demonstrates immunomodulatory potential and reduces HCC tumor growth when combined with ablative treatment. Purpose To evaluate the effect of incomplete cryoablation with or without MMP inhibition on the local immune response in residual tumors in a murine HCC model. Materials and Methods Sixty 8- to 10-week-old female BALB/c mice underwent HCC induction with use of orthotopic implantation of syngeneic Tib-75 cells. After 7 days, mice with a single lesion were randomized into treatment groups: (a) no treatment, (b) MMP inhibitor, (c) incomplete cryoablation, and (d) incomplete cryoablation and MMP inhibitor. Macrophage and T-cell subsets were assessed in tissue samples with use of immunohistochemistry and immunofluorescence (cell averages calculated using five 1-μm2 fields of view [FOVs]). C-X-C motif chemokine receptor type 3 (CXCR3)- and interferon γ (IFNγ)-positive T cells were assessed using flow cytometry. Groups were compared using unpaired Student t tests, one-way analysis of variance with Tukey correction, and the Kruskal-Wallis test with Dunn correction. Results Mice treated with incomplete cryoablation (n = 6) showed greater infiltration of CD206+ tumor-associated macrophages (mean, 1.52 cells per FOV vs 0.64 cells per FOV; P = .03) and MMP9-expressing cells (mean, 0.89 cells per FOV vs 0.11 cells per FOV; P = .03) compared with untreated controls (n = 6). Incomplete cryoablation with MMP inhibition (n = 6) versus without (n = 6) led to greater CD8+ T-cell (mean, 15.8% vs 8.29%; P = .04), CXCR3+CD8+ T-cell (mean, 11.64% vs 8.47%; P = .004), and IFNγ+CD8+ T-cell infiltration (mean, 11.58% vs 5.18%; P = .02). Conclusion In a mouse model of HCC, incomplete cryoablation and systemic MMP inhibition showed increased cytotoxic CD8+ T-cell infiltration into the residual tumor compared with either treatment alone. © RSNA, 2024 Supplemental material is available for this article. See also the editorial by Gemmete in this issue.
Conflict of interest statement
Figures

![Schematic overview of the study design. Approximately 2 million Tib-75
cells suspended in a biologically active matrix material were injected
subcapsularily into the liver of 8- to 10-week-old female BALB/c mice to
induce hepatocellular carcinoma (HCC). Mice were randomly divided into four
groups 7 days after tumor inoculation. Incomplete cryoablation (iCryo or
Cryo) was performed in groups 3 (n = 6) and 4 (n = 6) at day 0 (red
diamond). Groups 2 and 4 received intraperitoneal injection of
phosphate-buffered saline (PBS) (n = 6) or a broad-spectrum matrix
metalloproteinase (MMP) inhibitor diluted in phosphate-buffered saline (n =
6) every 2 days for a total of 7 doses (blue shading). Animals were killed
on day 17, and spleen and liver tissues were collected. Flow cytometry was
performed on tumor and tumor-adjacent tissue from four different groups:
control mice (no treatment), mice treated with the MMP inhibitor (n = 6),
mice treated with incomplete cryoablation (n = 6), and mice treated with
incomplete cryoablation and the MMP inhibitor (n = 6). Histopathologic
examination (immunohistochemistry, immunofluorescence, and hematoxylin and
eosin [H&E] staining) was performed on spleen and whole-liver tissue
from six different groups: the aforementioned four groups, in addition to
phosphate-buffered saline–treated mice (n = 6) and mice treated with
incomplete cryoablation and phosphate-buffered saline (n = 6).](https://cdn.ncbi.nlm.nih.gov/pmc/blobs/02e8/10902598/006cb394ebb8/radiol.232365.fig1.gif)



References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

