Impact of fosaprepitant in the prevention of nausea and emesis in head and neck cancer patients undergoing cisplatin-based chemoradiation: a pilot prospective study and a review of literature
- PMID: 38351333
- PMCID: PMC10942929
- DOI: 10.1007/s11547-024-01757-3
Impact of fosaprepitant in the prevention of nausea and emesis in head and neck cancer patients undergoing cisplatin-based chemoradiation: a pilot prospective study and a review of literature
Abstract
Purpose: Cisplatin-based chemoradiotherapy (CRT) is standard treatment for head and neck squamous cell carcinoma (HNSCC). However, IMRT may increase chemotherapy-induced nausea and vomiting (CINV). The purpose of this study is to investigate the effect of fosaprepitant in preventing CINV.
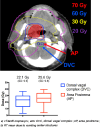
Methods: An infusion of 150 mg fosaprepitant was given through a 30 min. We assessed acute toxicity using CTCAE v.4 and the incidence of CINV using the FLIE questionnaire. The evaluation of CINV was done at the second and fifth weeks of CRT and 1 week after the end. The EORTC QLQ-HN 43 questionnaire was administered before treatment beginning (baseline), at second (T1) and fifth (T2) weeks. A dosimetric analysis was performed on dorsal nucleus of vagus (DVC) and area postrema (AP).
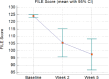
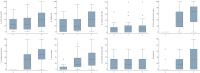
Results: Between March and November 2020, 24 patients were enrolled. No correlation was found between nausea and DVC mean dose (p = 0.573), and AP mean dose (p = 0.869). Based on the FLIE questionnaire, patients reported a mean score of 30.5 for nausea and 30 for vomiting during week 2 and 29.8 for nausea and 29.2 for vomiting during week 5. After treatment ended, the mean scores were 27.4 for nausea and 27.7 for vomiting. All patients completed the EORTC QLQ-HN 43. Significantly higher scores at T2 assessment than baseline were observed.
Conclusions: The use of fosaprepitant in preventing CINV reduced incidence of moderate to severe nausea and vomiting. No correlation has been found between nausea and median dose to DVC and AP.
Keywords: Chemoradiotherapy; Chemotherapy-induced nausea; Emesis; Fosaprepitant; Head and neck cancer.
© 2024. The Author(s).
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures
References
-
- Pignon JP, le Maître A, Maillard E, Bourhis J, MACH-NC Collaborative Group (2009) Meta-analysis of chemotherapy in head and neck cancer (MACH-NC): an update on 93 randomised trials and 17,346 patients. Radiother Oncol. 92(1):4–14 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

