Gut microbiota-derived trimethylamine N-Oxide: a novel target for the treatment of preeclampsia
- PMID: 38351748
- PMCID: PMC10868535
- DOI: 10.1080/19490976.2024.2311888
Gut microbiota-derived trimethylamine N-Oxide: a novel target for the treatment of preeclampsia
Abstract
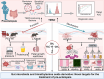
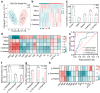
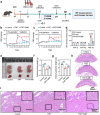
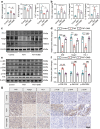
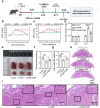
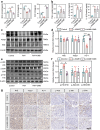
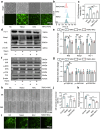
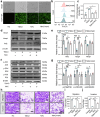
Pre-eclampsia (PE) is the most common complication of pregnancy and seriously threatens the health and safety of the mother and child. Studies have shown that an imbalance in gut microbiota can affect the progression of PE. Trimethylamine N-oxide (TMAO) is an intestinal microbiota-derived metabolite that is thought to be involved in the occurrence of PE; however, its causal relationship and mechanism remain unclear. In this clinical cohort study, including 28 patients with eclampsia and 39 matched healthy controls, fecal samples were collected for 16S rRNA gene sequencing, and serum was collected for targeted metabolomics research. The results showed that the level of TMAO and the abundance of its source bacteria had significantly increased in patients with PE, and were positively correlated with the clinical progression of PE. Fecal microbiota transplantation (FMT) was applied to an antibiotic-depleted-treated mouse model and targeted inhibition of TMAO. The results of the FMT experiment revealed that mice that received fecal microbiota transplantation from patients with PE developed typical PE symptoms and increased oxidative stress and inflammatory damage, both of which were reversed by 3,3-Dimethyl-1-butanol (DMB), a TMAO inhibitor, which also improved pregnancy outcomes in the model mice. Similar results were obtained in the classical NG-Nitroarginine methyl ester (L-NAME) induced PE mouse model. Mechanistically, TMAO promotes the progression of PE by regulating inflammatory and oxidative stress-related signaling pathways, affecting the migration and angiogenesis of vascular endothelial cells, as well as the migration and invasion of trophoblast cells. Our results reveal the role and mechanism of gut microbiota and TMAO in the progression of PE, provides new ideas for exploring the pathogenesis and therapeutic targets of PE, and determines the potential application value of TMAO as a target for PE intervention.
Keywords: Preeclampsia; TMAO; gut microbiota; placental trophoblast cells; vascular endothelial cells.
Conflict of interest statement
No potential conflict of interest was reported by the author(s).
Figures

References
-
- Brown MA, Magee LA, Kenny LC, Karumanchi SA, McCarthy FP, Saito S, Hall DR, Warren CE, Adoyi G, Ishaku S. Hypertensive disorders of pregnancy: ISSHP classification, diagnosis, and management recommendations for international practice. Hypertension. 2018;72(1):24–43. doi: 10.1161/HYPERTENSIONAHA.117.10803. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
