This is a preprint.
Focused Ultrasound Blood-Brain Barrier Opening Arrests the Growth and Formation of Cerebral Cavernous Malformations
- PMID: 38352349
- PMCID: PMC10862920
- DOI: 10.1101/2024.01.31.577810
Focused Ultrasound Blood-Brain Barrier Opening Arrests the Growth and Formation of Cerebral Cavernous Malformations
Update in
-
Focused ultrasound-microbubble treatment arrests the growth and formation of cerebral cavernous malformations.Nat Biomed Eng. 2025 Sep;9(9):1531-1546. doi: 10.1038/s41551-025-01390-z. Epub 2025 May 13. Nat Biomed Eng. 2025. PMID: 40360762 Free PMC article.
Abstract
Background: Cerebral cavernous malformations (CCM) are vascular lesions within the central nervous system, consisting of dilated and hemorrhage-prone capillaries. CCMs can cause debilitating neurological symptoms, and surgical excision or stereotactic radiosurgery are the only current treatment options. Meanwhile, transient blood-brain barrier opening (BBBO) with focused ultrasound (FUS) and microbubbles is now understood to exert potentially beneficial bioeffects, such as stimulation of neurogenesis and clearance of amyloid-β. Here, we tested whether FUS BBBO could be deployed therapeutically to control CCM formation and progression in a clinically-representative murine model.
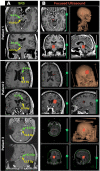
Methods: CCMs were induced in mice by postnatal, endothelial-specific Krit1 ablation. FUS was applied for BBBO with fixed peak-negative pressures (PNPs; 0.2-0.6 MPa) or passive cavitation detection-modulated PNPs. Magnetic resonance imaging (MRI) was used to target FUS treatments, evaluate safety, and measure longitudinal changes in CCM growth after BBBO.
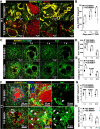
Results: FUS BBBO elicited gadolinium accumulation primarily at the perilesional boundaries of CCMs, rather than lesion cores. Passive cavitation detection and gadolinium contrast enhancement were comparable in CCM and wild-type mice, indicating that Krit1 ablation does not confer differential sensitivity to FUS BBBO. Acutely, CCMs exposed to FUS BBBO remained structurally stable, with no signs of hemorrhage. Longitudinal MRI revealed that FUS BBBO halted the growth of 94% of CCMs treated in the study. At 1 month, FUS BBBO-treated lesions lost, on average, 9% of their pre-sonication volume. In contrast, non-sonicated control lesions grew to 670% of their initial volume. Lesion control with FUS BBBO was accompanied by a marked reduction in the area and mesenchymal appearance of Krit mutant endothelium. Strikingly, in mice receiving multiple BBBO treatments with fixed PNPs, de novo CCM formation was significantly reduced by 81%. Mock treatment plans on MRIs of patients with surgically inaccessible lesions revealed their lesions are amenable to FUS BBBO with current clinical technology.
Conclusions: Our results establish FUS BBBO as a novel, non-invasive modality that can safely arrest murine CCM growth and prevent their de novo formation. As an incisionless, MR image-guided therapy with the ability to target eloquent brain locations, FUS BBBO offers an unparalleled potential to revolutionize the therapeutic experience and enhance the accessibility of treatments for CCM patients.
Figures


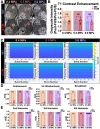
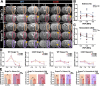
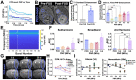

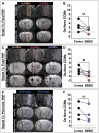
References
-
- Tanriover G, Sozen B, Seker A, Kilic T, Gunel M, Demir N. Ultrastructural analysis of vascular features in cerebral cavernous malformations. Clin Neurol Neurosurg [Internet]. 2013. [cited 2023 Jan 18];115:438–444. Available from: https://pubmed.ncbi.nlm.nih.gov/22776801/ - PubMed
-
- Wei S, Li Y, Polster SP, Shen L, Weber CR, Awad IA. Cerebral Cavernous Malformation Proteins in Barrier Maintenance and Regulation. Int J Mol Sci 2020, Vol 21, Page 675 [Internet]. 2020. [cited 2023 Jan 18];21:675. Available from: https://www.mdpi.com/1422-0067/21/2/675/htm - PMC - PubMed
-
- Zabramski JM, Wascher TM, Spetzler RF, Johnson B, Golfinos J, Drayer BP, Brown B, Rigamonti D, Brown G. The natural history of familial cavernous malformations: results of an ongoing study. J Neurosurg [Internet]. 1994. [cited 2022 Jul 29];80:422–432. Available from: https://thejns.org/view/journals/j-neurosurg/80/3/article-p422.xml - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
