Immunomodulation in non-alcoholic fatty liver disease: exploring mechanisms and applications
- PMID: 38352880
- PMCID: PMC10861763
- DOI: 10.3389/fimmu.2024.1336493
Immunomodulation in non-alcoholic fatty liver disease: exploring mechanisms and applications
Abstract
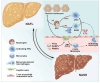
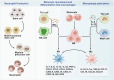
Non-alcoholic fatty liver disease (NAFLD) exhibits increased lipid enrichment in hepatocytes. The spectrum of this disease includes stages such as nonalcoholic simple fatty liver (NAFL), nonalcoholic steatohepatitis (NASH), and liver fibrosis. Changes in lifestyle behaviors have been a major factor contributing to the increased cases of NAFLD patients globally. Therefore, it is imperative to explore the pathogenesis of NAFLD, identify therapeutic targets, and develop new strategies to improve the clinical management of the disease. Immunoregulation is a strategy through which the organism recognizes and eliminates antigenic foreign bodies to maintain physiological homeostasis. In this process, multiple factors, including immune cells, signaling molecules, and cytokines, play a role in governing the evolution of NAFLD. This review seeks to encapsulate the advancements in research regarding immune regulation in NAFLD, spanning from underlying mechanisms to practical applications.
Keywords: Kupffer cells; NASH; immune cells; immunoregulation; non-alcoholic fatty liver disease.
Copyright © 2024 Guo, Wu, Xie, Wang and Lv.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

