Targeting oxidative phosphorylation to increase the efficacy of immune-combination therapy in renal cell carcinoma
- PMID: 38355278
- PMCID: PMC10868282
- DOI: 10.1136/jitc-2023-008226
Targeting oxidative phosphorylation to increase the efficacy of immune-combination therapy in renal cell carcinoma
Erratum in
-
Correction: Targeting oxidative phosphorylation to increase the efficacy of immune-combination therapy in renal cell carcinoma.J Immunother Cancer. 2024 Mar 5;12(3):e008226corr1. doi: 10.1136/jitc-2023-008226corr1. J Immunother Cancer. 2024. PMID: 38448039 Free PMC article. No abstract available.
Abstract
Background: Immune checkpoint inhibitors (ICIs) are the standard of care for metastatic renal cell carcinoma (RCC); however, most patients develop de novo or acquired resistance to ICIs. Oxidative phosphorylation (OXPHOS) has been rarely explored as a potential target for correcting ICI resistance.
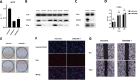
Methods: We systematically analyzed RNA sequencing and clinical data from CheckMate, JAVELIN Renal 101, and NCT01358721 clinical trials, and clinicopathological data of 25 patients from Tongji Hospital to investigate the relationship between OXPHOS and ICI resistance. The Ndufb8-knockdown Renca cell line was derived to determine the effect of OXPHOS on RCC immunotherapy in vivo.
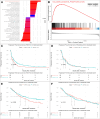
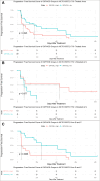
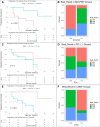
Results: An analysis of the CheckMate series data revealed that high OXPHOS levels are risk factors for ICI in patients with RCC, but are affected by thevon Hippel-Lindau protein (VHL) and hypoxia-inducible factor-1α status. This result is consistent with correlation between clinicopathological characteristics and prognostic observations at our institute. Knockdown of the mitochondrial complex I subunit Ndufb8 of the Renca cell line had no effect on cell growth and migration in vitro, but slowed down cell growth in vivo. Among anti-programmed death ligand 1 (PD-L1)-treated BALB/c mice, shNdufb8 Renca tumors grew slower than shControl Renca tumors and the corresponding mice survived longer. Flow cytometry revealed that CD8+ T cells in shNdufb8 Renca tumors, which were exposed to a lower degree of hypoxia and expressed less programmed death-1 (PD-1) and T-cell immunoglobulin domain and mucin domain 3 (TIM-3), secreted more interferon-γ after stimulation. Immunofluorescence demonstrated that the shNdufb8 Renca tumors had a higher proportion of CD8+ T cells and the proportion of these cells was lower in the hypoxic area.
Conclusions: OXPHOS is a reliable predictor of immunotherapy response in RCC and is more pronounced in metastatic lesions. RCC cells generate a hypoxic tumor microenvironment and inhibit T-cell function through oxidative metabolism, thereby leading to immunotherapy resistance.
Keywords: Immunotherapy; Metabolic Networks and Pathways; Renal Cell Carcinoma.
© Author(s) (or their employer(s)) 2024. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: No, there are no competing interests.
Figures

References
-
- Powles T, Plimack ER, Soulières D, et al. Pembrolizumab plus axitinib versus sunitinib monotherapy as first-line treatment of advanced renal cell carcinoma (KEYNOTE-426): extended follow-up from a randomised, open-label, phase 3 trial. The Lancet Oncology 2020;21:1563–73. 10.1016/S1470-2045(20)30436-8 - DOI - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Research Materials
