Targeting solid tumor antigens with chimeric receptors: cancer biology meets synthetic immunology
- PMID: 38355356
- PMCID: PMC11006585
- DOI: 10.1016/j.trecan.2024.01.003
Targeting solid tumor antigens with chimeric receptors: cancer biology meets synthetic immunology
Abstract
Chimeric antigen receptor (CAR) T cell therapy is a medical breakthrough in the treatment of B cell malignancies. There is intensive focus on developing solid tumor-targeted CAR-T cell therapies. Although clinically approved CAR-T cell therapies target B cell lineage antigens, solid tumor targets include neoantigens and tumor-associated antigens (TAAs) with diverse roles in tumor biology. Multiple early-stage clinical trials now report encouraging signs of efficacy for CAR-T cell therapies that target solid tumors. We review the landscape of solid tumor target antigens from the perspective of cancer biology and gene regulation, together with emerging clinical data for CAR-T cells targeting these antigens. We then discuss emerging synthetic biology strategies and their application in the clinical development of novel cellular immunotherapies.
Keywords: CAR-T cells; antigen targeting; cellular immunotherapy; solid tumor; synthetic biology.
Copyright © 2024 Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests G.J.K and J.Y.K declare no competing interests. M.V.M. is an inventor on patents related to adoptive cell therapies held by Massachusetts General Hospital (some licensed to Promab) and the University of Pennsylvania (some licensed to Novartis). M.V.M. holds equity in 2SeventyBio, Century Therapeutics, Neximmune, Oncternal, and TCR2, and has served as a consultant for multiple companies involved in cell therapies. M.V.M. is a member of the Board of Directors of 2SeventyBio. M.J. is an inventor on patents related to targeted protein degradation and adoptive cell therapies held by Massachusetts General Hospital and the Broad Institute of MIT and Harvard. M.J. has received consulting fees from RA Ventures. M.J. has an immediate family member who is a founder and shareholder of Orna Therapeutics, a shareholder of Myeloid Therapeutics, and a founder, employee, shareholder, and member of the scientific advisory board of Ganna Therapeutics.
Figures
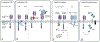

References
-
- Majzner RG et al. (2020) CD58 Aberrations Limit Durable Responses to CD19 CAR in Large B Cell Lymphoma Patients Treated with Axicabtagene Ciloleucel but Can be Overcome through Novel CAR Engineering. Blood 136, 53–54
-
- Hou AJ et al. (2021) Navigating CAR-T cells through the solid-tumour microenvironment. Nat. Rev. Drug Discov 20, 531–550 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

