The Temozolomide-Doxorubicin paradox in Glioblastoma in vitro-in silico preclinical drug-screening
- PMID: 38355655
- PMCID: PMC10866941
- DOI: 10.1038/s41598-024-53684-y
The Temozolomide-Doxorubicin paradox in Glioblastoma in vitro-in silico preclinical drug-screening
Abstract
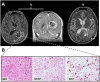
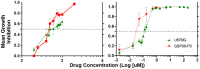
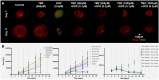
Adjuvant Temozolomide is considered the front-line Glioblastoma chemotherapeutic treatment; yet not all patients respond. Latest trends in clinical trials usually refer to Doxorubicin; yet it can lead to severe side-effects if administered in high doses. While Glioblastoma prognosis remains poor, little is known about the combination of the two chemotherapeutics. Patient-derived spheroids were generated and treated with a range of Temozolomide/Doxorubicin concentrations either as monotherapy or in combination. Optical microscopy was used to monitor the growth pattern and cell death. Based on the monotherapy experiments, we developed a probabilistic mathematical framework in order to describe the drug-induced effect at the single-cell level and simulate drug doses in combination assuming probabilistic independence. Doxorubicin was found to be effective in doses even four orders of magnitude less than Temozolomide in monotherapy. The combination therapy doses tested in vitro were able to lead to irreversible growth inhibition at doses where monotherapy resulted in relapse. In our simulations, we assumed both drugs are anti-mitotic; Temozolomide has a growth-arrest effect, while Doxorubicin is able to cumulatively cause necrosis. Interestingly, under no mechanistic synergy assumption, the in silico predictions underestimate the in vitro results. In silico models allow the exploration of a variety of potential underlying hypotheses. The simulated-biological discrepancy at certain doses indicates a supra-additive response when both drugs are combined. Our results suggest a Temozolomide-Doxorubicin dual chemotherapeutic scheme to both disable proliferation and increase cytotoxicity against Glioblastoma.
Keywords: Brain cancer; Computational models; Doxorubicin; Preclinical drug-screening; Temozolomide.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures

Similar articles
-
The effect of temozolomide in combination with doxorubicin in glioblastoma cells in vitro.J Immunoassay Immunochem. 2020 Nov 1;41(6):1033-1043. doi: 10.1080/15321819.2020.1819309. Epub 2020 Oct 6. J Immunoassay Immunochem. 2020. PMID: 33021886
-
Synergistic Effects of Temozolomide and Doxorubicin in the Treatment of Glioblastoma Multiforme: Enhancing Efficacy through Combination Therapy.Molecules. 2024 Feb 14;29(4):840. doi: 10.3390/molecules29040840. Molecules. 2024. PMID: 38398592 Free PMC article.
-
The Combined Treatment with Chemotherapeutic Agents and the Dualsteric Muscarinic Agonist Iper-8-Naphthalimide Affects Drug Resistance in Glioblastoma Stem Cells.Cells. 2021 Jul 24;10(8):1877. doi: 10.3390/cells10081877. Cells. 2021. PMID: 34440646 Free PMC article.
-
Involvement of Intracellular Cholesterol in Temozolomide-Induced Glioblastoma Cell Death.Neurol Med Chir (Tokyo). 2018 Jul 15;58(7):296-302. doi: 10.2176/nmc.ra.2018-0040. Epub 2018 Jun 13. Neurol Med Chir (Tokyo). 2018. PMID: 29899179 Free PMC article. Review.
-
Is Autophagy Inhibition in Combination with Temozolomide a Therapeutically Viable Strategy?Cells. 2023 Feb 7;12(4):535. doi: 10.3390/cells12040535. Cells. 2023. PMID: 36831202 Free PMC article. Review.
Cited by
-
Tinostamustine (EDO-S101) and Its Combination with Celecoxib or Temozolomide as a Therapeutic Option for Adult-Type Diffuse Gliomas.Int J Mol Sci. 2025 Jan 14;26(2):661. doi: 10.3390/ijms26020661. Int J Mol Sci. 2025. PMID: 39859375 Free PMC article.
-
Enhancing Therapeutic Approaches in Glioblastoma with Pro-Oxidant Treatments and Synergistic Combinations: In Vitro Experience of Doxorubicin and Photodynamic Therapy.Int J Mol Sci. 2024 Jul 9;25(14):7525. doi: 10.3390/ijms25147525. Int J Mol Sci. 2024. PMID: 39062770 Free PMC article.
-
Current Non-Metal Nanoparticle-Based Therapeutic Approaches for Glioblastoma Treatment.Biomedicines. 2024 Aug 11;12(8):1822. doi: 10.3390/biomedicines12081822. Biomedicines. 2024. PMID: 39200286 Free PMC article. Review.
References
-
- Villodre ES, Kipper FC, Silva AO, Lenz G, Lopez PLdC. Low dose of doxorubicin potentiates the effect of Temozolomide in glioblastoma cells. Mol. Neurobiol. 2018;55(5):4185–4194. - PubMed
-
- MacDiarmid JA, Langova V, Bailey D, Pattison ST, Pattison SL, Christensen N, et al. Targeted doxorubicin delivery to brain tumors via minicells: Proof of principle using dogs with spontaneously occurring tumors as a model. PLoS ONE. 2016;11(4):e0151832. doi: 10.1371/journal.pone.0151832. - DOI - PMC - PubMed
-
- Ananda S, Nowak AK, Cher L, Dowling AJ, Brown C, Simes RJ, et al. Phase II trial of combined temozolomide and pegylated liposomal doxorubicin in the treatment of patients with glioblastoma multiforme following concurrent radiotherapy and chemotherapy. J. Clin. Oncol. 2010;28(15_suppl):2072. doi: 10.1200/jco.2010.28.15_suppl.2072. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

