SARS-CoV-2 spike protein-ACE2 interaction increases carbohydrate sulfotransferases and reduces N-acetylgalactosamine-4-sulfatase by p38 MAPK
- PMID: 38355690
- PMCID: PMC10866996
- DOI: 10.1038/s41392-024-01741-3
SARS-CoV-2 spike protein-ACE2 interaction increases carbohydrate sulfotransferases and reduces N-acetylgalactosamine-4-sulfatase by p38 MAPK
Abstract
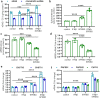
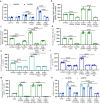
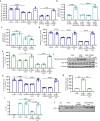
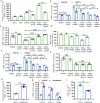
Immunostaining in lungs of patients who died with COVID-19 infection showed increased intensity and distribution of chondroitin sulfate and decline in N-acetylgalactostamine-4-sulfatase (Arylsulfatase B; ARSB). To explain these findings, human small airway epithelial cells were exposed to the SARS-CoV-2 spike protein receptor binding domain (SPRBD) and transcriptional mechanisms were investigated. Phospho-p38 MAPK and phospho-SMAD3 increased following exposure to the SPRBD, and their inhibition suppressed the promoter activation of the carbohydrate sulfotransferases CHST15 and CHST11, which contributed to chondroitin sulfate biosynthesis. Decline in ARSB was mediated by phospho-38 MAPK-induced N-terminal Rb phosphorylation and an associated increase in Rb-E2F1 binding and decline in E2F1 binding to the ARSB promoter. The increases in chondroitin sulfotransferases were inhibited when treated with phospho-p38-MAPK inhibitors, SMAD3 (SIS3) inhibitors, as well as antihistamine desloratadine and antibiotic monensin. In the mouse model of carrageenan-induced systemic inflammation, increases in phospho-p38 MAPK and expression of CHST15 and CHST11 and declines in DNA-E2F binding and ARSB expression occurred in the lung, similar to the observed effects in this SPRBD model of COVID-19 infection. Since accumulation of chondroitin sulfates is associated with fibrotic lung conditions and diffuse alveolar damage, increased attention to p38-MAPK inhibition may be beneficial in ameliorating Covid-19 infections.
© 2024. This is a U.S. Government work and not under copyright protection in the US; foreign copyright protection may apply.
Conflict of interest statement
The authors declare no competing interests.
Figures
Similar articles
-
Increase in Chondroitin Sulfate and Decline in Arylsulfatase B May Contribute to Pathophysiology of COVID-19 Respiratory Failure.Pathobiology. 2022;89(2):81-91. doi: 10.1159/000519542. Epub 2021 Nov 17. Pathobiology. 2022. PMID: 34788765
-
Inhibition of Phosphatase Activity Follows Decline in Sulfatase Activity and Leads to Transcriptional Effects through Sustained Phosphorylation of Transcription Factor MITF.PLoS One. 2016 Apr 14;11(4):e0153463. doi: 10.1371/journal.pone.0153463. eCollection 2016. PLoS One. 2016. PMID: 27078017 Free PMC article.
-
Arylsulfatase B induces melanoma apoptosis by the ubiquitin ligase COP1.J Biol Chem. 2025 Jun 23;301(8):110402. doi: 10.1016/j.jbc.2025.110402. Online ahead of print. J Biol Chem. 2025. PMID: 40562100 Free PMC article.
-
Antibody tests for identification of current and past infection with SARS-CoV-2.Cochrane Database Syst Rev. 2022 Nov 17;11(11):CD013652. doi: 10.1002/14651858.CD013652.pub2. Cochrane Database Syst Rev. 2022. PMID: 36394900 Free PMC article.
-
Rapid, point-of-care antigen tests for diagnosis of SARS-CoV-2 infection.Cochrane Database Syst Rev. 2022 Jul 22;7(7):CD013705. doi: 10.1002/14651858.CD013705.pub3. Cochrane Database Syst Rev. 2022. PMID: 35866452 Free PMC article.
Cited by
-
Reprogramming the myocardial infarction microenvironment with melanin-based composite nanomedicines in mice.Nat Commun. 2024 Aug 6;15(1):6651. doi: 10.1038/s41467-024-50854-4. Nat Commun. 2024. PMID: 39103330 Free PMC article.
References
-
- Venkatesan N, Ouzzine M, Kolb M, Netter P, Ludwig MS. Increased deposition of chondroitin/dermatan sulfate glycosaminoglycan and upregulation of β1,3- glucuronosyltransferase I in pulmonary fibrosis. Am. J. Physiol. Lung Cell Mol. Physiol. 2011;300:L191–L203. doi: 10.1152/ajplung.00214.2010. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

