Orientational Behavior and Vibrational Response of Glycine at Aqueous Interfaces
- PMID: 38358315
- PMCID: PMC10895693
- DOI: 10.1021/acs.jpclett.3c02930
Orientational Behavior and Vibrational Response of Glycine at Aqueous Interfaces
Abstract
Aqueous glycine plays many different roles in living systems, from being a building block for proteins to being a neurotransmitter. To better understand its fundamental behavior, we study glycine's orientational behavior near model aqueous interfaces, in the absence and presence of electric fields and biorelevant ions. To this purpose, we use a surface-specific technique called heterodyne-detected vibrational sum-frequency generation spectroscopy (HD-VSFG). Using HD-VSFG, we directly probe the symmetric and antisymmetric stretching vibrations of the carboxylate group of zwitterionic glycine. From their relative amplitudes, we infer the zwitterion's orientation near surfactant-covered interfaces and find that it is governed by both electrostatic and surfactant-specific interactions. By introducing additional ions, we observe that the net orientation is altered by the enhanced ionic strength, indicating a change in the balance of the electrostatic and surfactant-specific interactions.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
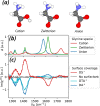
 . In the presented spectra, we subtracted
the infrared absorption of the solvent and normalized the signals
to the sample thickness, see Supporting Information. The main vibrational features are assigned in Table 1. (c) HD-VSFG spectra (Im(χ(2)), SSP polarization) of 1 M
glycine solutions at neutral pD, at the neat D2O/air interface
and in the presence of monolayers of charged surfactants. The above
spectra are presented after subtracting the corresponding HD-VSFG
spectra of neat and surfactant-covered D2O/air interfaces,
see Figure S5.
. In the presented spectra, we subtracted
the infrared absorption of the solvent and normalized the signals
to the sample thickness, see Supporting Information. The main vibrational features are assigned in Table 1. (c) HD-VSFG spectra (Im(χ(2)), SSP polarization) of 1 M
glycine solutions at neutral pD, at the neat D2O/air interface
and in the presence of monolayers of charged surfactants. The above
spectra are presented after subtracting the corresponding HD-VSFG
spectra of neat and surfactant-covered D2O/air interfaces,
see Figure S5.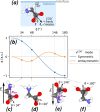
 of the two main carboxylate modes of zwitterionic
glycine, derived using the theoretical and experimental results of
earlier works.− These works use the assumption that the COO– group
can freely rotate around the C–C bond. We show
of the two main carboxylate modes of zwitterionic
glycine, derived using the theoretical and experimental results of
earlier works.− These works use the assumption that the COO– group
can freely rotate around the C–C bond. We show  ratios in Table 2. Below: Illustration of zwitterionic glycine
molecules, oriented due to (c) the electric field induced by negative
surface charges, (d) the interaction of the amine group and the surfactant
monolayer, (e) the electric field induced by positive surface charges,
and (f) the interaction of the carboxylate group and the surfactant
monolayer.
ratios in Table 2. Below: Illustration of zwitterionic glycine
molecules, oriented due to (c) the electric field induced by negative
surface charges, (d) the interaction of the amine group and the surfactant
monolayer, (e) the electric field induced by positive surface charges,
and (f) the interaction of the carboxylate group and the surfactant
monolayer.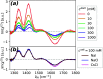
 of zwitterionic glycine as well as that
of the
of zwitterionic glycine as well as that
of the  bending mode of H2O (ν̃IR = 1643 cm–1). (b) Effect of exchanging salt ions (csalt = 100 mM). For both sets of spectra, we subtracted the quadrupolar
SFG contribution of neat H2O, which does not change with
added salt concentration.
bending mode of H2O (ν̃IR = 1643 cm–1). (b) Effect of exchanging salt ions (csalt = 100 mM). For both sets of spectra, we subtracted the quadrupolar
SFG contribution of neat H2O, which does not change with
added salt concentration.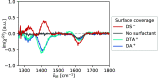
Similar articles
-
Direct Observation of the Orientation of Urea Molecules at Charged Interfaces.J Phys Chem Lett. 2021 Nov 11;12(44):10823-10828. doi: 10.1021/acs.jpclett.1c03012. Epub 2021 Nov 2. J Phys Chem Lett. 2021. PMID: 34726406 Free PMC article.
-
Orientation of Methylguanidinium Ions at the Water-Air Interface.J Phys Chem C Nanomater Interfaces. 2017 Oct 26;121(42):23398-23405. doi: 10.1021/acs.jpcc.7b03752. Epub 2017 Sep 14. J Phys Chem C Nanomater Interfaces. 2017. PMID: 29129985 Free PMC article.
-
Theoretical vibrational sum-frequency generation spectroscopy of water near lipid and surfactant monolayer interfaces.J Chem Phys. 2014 Nov 14;141(18):18C502. doi: 10.1063/1.4895546. J Chem Phys. 2014. PMID: 25399167
-
Molecular orientation of small carboxylates at the water-air interface.Phys Chem Chem Phys. 2022 May 4;24(17):10134-10139. doi: 10.1039/d1cp05471f. Phys Chem Chem Phys. 2022. PMID: 35416809
-
Environmental chemistry at vapor/water interfaces: insights from vibrational sum frequency generation spectroscopy.Annu Rev Phys Chem. 2012;63:107-30. doi: 10.1146/annurev-physchem-032511-143811. Epub 2012 Jan 3. Annu Rev Phys Chem. 2012. PMID: 22224702 Review.
Cited by
-
Observation of Electrostatically Driven Surface Adsorption in Mixed Surfactant Systems.J Phys Chem Lett. 2024 Feb 15;15(6):1596-1602. doi: 10.1021/acs.jpclett.3c03377. Epub 2024 Feb 2. J Phys Chem Lett. 2024. PMID: 38306467 Free PMC article.
References
LinkOut - more resources
Full Text Sources

