Enantioselective nickel-catalyzed Mizoroki-Heck cyclizations of amide electrophiles
- PMID: 38362425
- PMCID: PMC10866352
- DOI: 10.1039/d3sc05797f
Enantioselective nickel-catalyzed Mizoroki-Heck cyclizations of amide electrophiles
Abstract
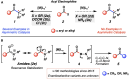
Amide cross-couplings that rely on C-N bond activation by transition metal catalysts have emerged as valuable synthetic tools. Despite numerous discoveries in this field, no catalytic asymmetric variants have been disclosed to date. Herein, we demonstrate the first such transformation, which is the Mizoroki-Heck cyclization of amide substrates using asymmetric nickel catalysis. This proof-of-concept study provides an entryway to complex enantioenriched polycyclic scaffolds and advances the field of amide C-N bond activation chemistry.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures







Similar articles
-
Mizoroki-Heck Cyclizations of Amide Derivatives for the Introduction of Quaternary Centers.Angew Chem Int Ed Engl. 2017 Jun 1;56(23):6567-6571. doi: 10.1002/anie.201703174. Epub 2017 May 3. Angew Chem Int Ed Engl. 2017. PMID: 28467029 Free PMC article.
-
Nickel-Catalyzed Ligand-Controlled Selective Reductive Cyclization/Cross-Couplings.Acc Chem Res. 2023 Mar 7;56(5):515-535. doi: 10.1021/acs.accounts.2c00771. Epub 2023 Jan 23. Acc Chem Res. 2023. PMID: 36688822
-
Transition-Metal (Pd, Ni, Mn)-Catalyzed C-C Bond Constructions Involving Unactivated Alkyl Halides and Fundamental Synthetic Building Blocks.Acc Chem Res. 2019 Apr 16;52(4):1134-1144. doi: 10.1021/acs.accounts.9b00044. Epub 2019 Mar 25. Acc Chem Res. 2019. PMID: 30908013 Free PMC article.
-
Nickel-catalyzed reductive cyclizations and couplings.Angew Chem Int Ed Engl. 2004 Jul 26;43(30):3890-908. doi: 10.1002/anie.200300634. Angew Chem Int Ed Engl. 2004. PMID: 15274210 Review.
-
Nickel-Catalyzed Arylative Cyclizations of Alkyne- and Allene-Tethered Electrophiles using Arylboron Reagents.Chemistry. 2022 Mar 28;28(18):e202104230. doi: 10.1002/chem.202104230. Epub 2022 Jan 27. Chemistry. 2022. PMID: 34986277 Free PMC article. Review.
Cited by
-
Mechanistic insights on C(acyl)-N functionalisation mediated by late transition metals.Dalton Trans. 2024 Dec 3;53(47):18803-18818. doi: 10.1039/d4dt01829j. Dalton Trans. 2024. PMID: 39115156 Review.
References
-
- Johansson Seechurn C. Kitching M. O. Colacot T. J. Snieckus V. Angew. Chem., Int. Ed. 2012;51:5062–5085. doi: 10.1002/anie.201107017. - DOI - PubMed
- Hassan J. Sevignon M. Gozzi C. Schulz E. Lemaire M. Chem. Rev. 2002;102:1359–1470. doi: 10.1021/cr000664r. - DOI - PubMed
- Jiang L. and Buchwald S. L., in Metal-Catalyzed Cross- Coupling Reactions, 2nd edn, ed. A. Meijere and F. Diederich, Wiley VCH, Weinheim, 2004, pp. 699−760
- Corbet J. P. Mignani G. Chem. Rev. 2006;106:2651–2710. doi: 10.1021/cr0505268. - DOI - PubMed
- Negishi E. Bull. Chem. Soc. Jpn. 2007;80:233–257. doi: 10.1246/bcsj.80.233. - DOI
- Shen H. C., in Application of Transition Metal Catalysis in Drug Discovery and Development: An Industrial Perspective, ed. M. L. Crawley and B. M. Trost, Wiley, Hoboken, 2012, pp. 25−96
- Campeau L. C. Hazari N. Organometallics. 2019;38:3–35. doi: 10.1021/acs.organomet.8b00720. - DOI - PMC - PubMed
- Suzuki A. Angew. Chem., Int. Ed. 2011;50:6722–6737. doi: 10.1002/anie.201101379. - DOI - PubMed
- Negishi E. Angew. Chem., Int. Ed. 2011;50:6738–6764. doi: 10.1002/anie.201101380. - DOI - PubMed
-
-
For select reviews on C–O, C–N and C–F bond cleaving reactions, see:
- Boit T. B. Bulger A. S. Dander J. E. Garg N. K. ACS Catal. 2020;10:12109–12126. doi: 10.1021/acscatal.0c03334. - DOI - PMC - PubMed
- Zhang S. Q. Hong X. Acc. Chem. Res. 2021;54:2158–2171. doi: 10.1021/acs.accounts.1c00050. - DOI - PubMed
- Baviskar B. A. Ajmire P. V. Chumbhale D. S. Khan M. S. Kuchake V. G. Singupuram M. Laddha P. R. Sustainable Chem. Pharm. 2023;32:100953. doi: 10.1016/j.scp.2022.100953. - DOI
- Zeng X. Huang X. Adv. Anal. Chem. 2022;12:68–75. doi: 10.12677/AAC.2022.122010. - DOI
- Tian M. Liu M. Pure Appl. Chem. 2021;93:799–810. doi: 10.1515/pac-2021-0110. - DOI
- Cornella J. Zarate C. Martin R. Chem. Soc. Rev. 2014;43:8081–8097. doi: 10.1039/C4CS00206G. - DOI - PubMed
- Tobisu M. Chatani N. Acc. Chem. Res. 2015;48:1717–1726. doi: 10.1021/acs.accounts.5b00051. - DOI - PubMed
- Liu F. Jiang H. J. Zhou Y. Shi Z. Chin. J. Chem. 2020;38:855–863. doi: 10.1002/cjoc.201900506. - DOI
- Zhou T. Szostak M. Catal. Sci. Technol. 2020;10:5702–5739. doi: 10.1039/D0CY01159B. - DOI - PMC - PubMed
- Qiu Z. Li C. J. Chem. Rev. 2020;120:10454–10515. doi: 10.1021/acs.chemrev.0c00088. - DOI - PubMed
- Ahrens T. Kohlmann J. Ahrens M. Braun T. Chem. Rev. 2015;115:931–972. doi: 10.1021/cr500257c. - DOI - PubMed
- Fu L. Chen Q. Nishihara Y. Chem. Rec. 2021;21:3394–3410. doi: 10.1002/tcr.202100053. - DOI - PubMed
- Becica J. Leitch D. C. Synlett. 2021;32:641–646. doi: 10.1055/a-1306-3228. - DOI
-
-
-
For select reviews on acyl electrophiles see:
- Buchspies J. Szostak M. Catal. 2019;9:53.
- Takise R. Muto K. Yamaguchi J. Chem. Soc. Rev. 2017;46:5864–5888. doi: 10.1039/C7CS00182G. - DOI - PubMed
- Zhou T. Szostak M. Catal. Sci. Technol. 2020;10:5702–5739. doi: 10.1039/D0CY01159B. - DOI - PMC - PubMed
- Gooßen L. J. Rodríguez N. Gooßen K. Angew. Chem., Int. Ed. 2008;47:3100–3120. doi: 10.1002/anie.200704782. - DOI - PubMed
- Dander J. E. Garg N. K. ACS Catal. 2017;7:1413–1423. doi: 10.1021/acscatal.6b03277. - DOI - PMC - PubMed
- Li G. Ma S. Szostak M. Trends Chem. 2020;2:914–928. doi: 10.1016/j.trechm.2020.08.001. - DOI
-
-
-
For this context, “conventional” is used to refer to cross-couplings employing a nucleophile and electrophile. For select studies on asymmetric transition-metal-catalyzed reactions using thioester electrophiles, see:
- Banchini F. Leroux B. Le Gall E. Presset M. Jackowski O. Chemla F. Perez-Luna A. Chem.–Eur. J. 2023:e202301084. doi: 10.1002/chem.202301084. - DOI - PubMed
- Oost R. Misale A. Maulide N. Angew. Chem., Int. Ed. 2016;55:4587–4590. doi: 10.1002/anie.201600597. - DOI - PubMed
- Liu M. Wang X. Guo Z. Li H. Huang W. Xu H. Dai H.-X. Org. Lett. 2021;23:6299–6304. doi: 10.1021/acs.orglett.1c02093. - DOI - PubMed
-
-
-
For select studies on asymmetric transition-metal-catalyzed reductive or photochemical cross-coupling reactions using anhydride or acid chloride electrophiles, see:
- Cherney A. H. Kadunce N. T. Reisman S. E. J. Am. Chem. Soc. 2013;135:7442–7445. doi: 10.1021/ja402922w. - DOI - PubMed
- Ji H. Lin D. Tai L. Li X. Shi Y. Han Q. Chen L.-A. J. Am. Chem. Soc. 2022;144:23019–23029. doi: 10.1021/jacs.2c10072. - DOI - PubMed
- Gandolfo E. Tang X. Roy S. R. Melchiorre P. Angew. Chem. 2019;131:17010–17014. doi: 10.1002/ange.201910168. - DOI - PMC - PubMed
-

