Integrating hydrogen utilization in CO2 electrolysis with reduced energy loss
- PMID: 38365776
- PMCID: PMC10873292
- DOI: 10.1038/s41467-024-45787-x
Integrating hydrogen utilization in CO2 electrolysis with reduced energy loss
Abstract
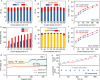
Electrochemical carbon dioxide reduction reaction using sustainable energy is a promising approach of synthesizing chemicals and fuels, yet is highly energy intensive. The oxygen evolution reaction is particularly problematic, which is kinetically sluggish and causes anodic carbon loss. In this context, we couple CO2 electrolysis with hydrogen oxidation reaction in a single electrochemical cell. A Ni(OH)2/NiOOH mediator is used to fully suppress the anodic carbon loss and hydrogen oxidation catalyst poisoning by migrated reaction products. This cell is highly flexible in producing either gaseous (CO) or soluble (formate) products with high selectivity (up to 95.3%) and stability (>100 h) at voltages below 0.9 V (50 mA cm-2). Importantly, thanks to the "transferred" oxygen evolution reaction to a water electrolyzer with thermodynamically and kinetically favored reaction conditions, the total polarization loss and energy consumption of our H2-integrated CO2 reduction reaction, including those for hydrogen generation, are reduced up to 22% and 42%, respectively. This work demonstrates the opportunity of combining CO2 electrolysis with the hydrogen economy, paving the way to the possible integration of various emerging energy conversion and storage approaches for improved energy/cost effectiveness.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures




References
-
- De Luna P, et al. What would it take for renewably powered electrosynthesis to displace petrochemical processes? Science. 2019;364:350. - PubMed
-
- Zhi, W. Y. et al. Efficient electroreduction of CO2 to C2-C3 products on Cu/Cu2O@N-doped graphene. J. CO2Util.50, 101594 (2021).
-
- Ma W, et al. Electrocatalytic reduction of CO2 to ethylene and ethanol through hydrogen-assisted C–C coupling over fluorine-modified copper. Nat. Catal. 2020;3:478–487. doi: 10.1038/s41929-020-0450-0. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources

