Histochemical, metabolic and ultrastructural changes in leaf patelliform nectaries explain extrafloral nectar synthesis and secretion in Clerodendrum chinense
- PMID: 38366151
- PMCID: PMC11037555
- DOI: 10.1093/aob/mcae019
Histochemical, metabolic and ultrastructural changes in leaf patelliform nectaries explain extrafloral nectar synthesis and secretion in Clerodendrum chinense
Abstract
Background and aims: Extrafloral nectaries are nectar-secreting structures present on vegetative parts of plants which provide indirect defences against herbivore attack. Extrafloral nectaries in Clerodendrum chinense are patelliform-shaped specialized trichomatous structures. However, a complete understanding of patelliform extrafloral nectaries in general, and of C. chinense in particular, has not yet been established to provide fundamental insight into the cellular physiological machinery involved in nectar biosynthesis and secretory processes.
Methods: We studied temporal changes in the morphological, anatomical and ultrastructural features in the architectures of extrafloral nectaries. We also compared metabolite profiles of extrafloral nectar, nectary tissue, non-nectary tissue and phloem sap. Further, both in situ histolocalization and normal in vitro activities of enzymes related to sugar metabolism were examined.
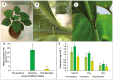
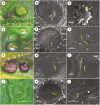
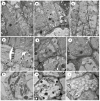
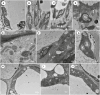
Key results: Four distinct tissue regions in the nectar gland were revealed from histochemical characterization, among which the middle nectariferous tissue was found to be the metabolically active region, while the intermediate layer was found to be lipid-rich. Ultrastructural study showed the presence of a large number of mitochondria along with starch-bearing chloroplasts in the nectariferous region. However, starch depletion was noted with progressive maturation of nectaries. Metabolite analysis revealed compositional differences among nectar, phloem sap, nectary and non-nectary tissue. Invertase activity was higher in secretory stages and localized in nectariferous tissue and adjacent region.
Conclusions: Our study suggests extrafloral nectar secretion in C. chinense to be both eccrine and merocrine in nature. A distinct intermediate lipid-rich layer that separates the epidermis from nectary parenchyma was revealed, which possibly acts as a barrier to water flow in nectar. This study also revealed a distinction between nectar and phloem sap, and starch could act as a nectar precursor, as evidenced from enzymatic and ultrastructural studies. Thus, our findings on changing architecture of extrafloral nectaries with temporal secretion revealed a cell physiological process involved in nectar biosynthesis and secretion.
Keywords: Clerodendrum chinense; in situ histolocalization; Extrafloral nectary; nectar; nectariferous tissue; patelliform nectaries; phloem sap.
© The Author(s) 2024. Published by Oxford University Press on behalf of the Annals of Botany Company. All rights reserved. For commercial re-use, please contact reprints@oup.com for reprints and translation rights for reprints. All other permissions can be obtained through our RightsLink service via the Permissions link on the article page on our site—for further information please contact journals.permissions@oup.com.
Figures



Similar articles
-
Deciphering histochemical and ultrastructural features of calyx nectaries to understand the secretory process in two species of Clerodendrum (Lamiaceae).Protoplasma. 2025 May 28. doi: 10.1007/s00709-025-02073-x. Online ahead of print. Protoplasma. 2025. PMID: 40437262
-
Morphological and anatomical traits during development: Highlighting extrafloral nectaries in Passiflora organensis.Microsc Res Tech. 2022 Aug;85(8):2784-2794. doi: 10.1002/jemt.24127. Epub 2022 Apr 14. Microsc Res Tech. 2022. PMID: 35421272
-
Extrafloral nectaries of four varieties of Chamaecrista ramosa (Vogel) H.S.Irwin & Barneby (Fabaceae): anatomy, chemical nature, mechanisms of nectar secretion, and elimination.Protoplasma. 2018 Nov;255(6):1635-1647. doi: 10.1007/s00709-018-1253-x. Epub 2018 Apr 27. Protoplasma. 2018. PMID: 29704049
-
Nightshade Wound Secretion: The World's Simplest Extrafloral Nectar?Trends Plant Sci. 2016 Aug;21(8):637-638. doi: 10.1016/j.tplants.2016.06.004. Trends Plant Sci. 2016. PMID: 27375224 Review.
-
How common is the ability of extrafloral nectaries to produce nectar droplets, to secrete nectar during the night and to store starch?Plant Biol (Stuttg). 2012 Sep;14(5):691-5. doi: 10.1111/j.1438-8677.2012.00616.x. Epub 2012 Jun 7. Plant Biol (Stuttg). 2012. PMID: 22672217 Review.
Cited by
-
Deciphering histochemical and ultrastructural features of calyx nectaries to understand the secretory process in two species of Clerodendrum (Lamiaceae).Protoplasma. 2025 May 28. doi: 10.1007/s00709-025-02073-x. Online ahead of print. Protoplasma. 2025. PMID: 40437262
-
Use of polyethylene glycol as an alternative to optimal cutting temperature medium in freeze sectioning for plant histochemical studies.Protoplasma. 2025 May;262(3):721-737. doi: 10.1007/s00709-024-02008-y. Epub 2024 Dec 18. Protoplasma. 2025. PMID: 39692865
-
Histochemical and molecular analyses reveal an insight into the scent volatiles synthesis and emission in ephemeral flowers of Murraya paniculata (L.) Jack.Planta. 2024 Oct 18;260(5):119. doi: 10.1007/s00425-024-04552-6. Planta. 2024. PMID: 39422757
References
-
- Afzal S, Singh NK, Singh N, Chaudhury N.. 2021. Structural analysis of extrafloral nectaries of Senna occidentalis L.: insights on diversity and evolution. Planta 254: 125. - PubMed
-
- Beccari N, Mazzi V.. 1966. Manuale di tecnica microscopica. Guida pratica alla ricercar istologica e istochimica. Torino: Società Editrice Libraria.
-
- Bentley BL. 1977. Extrafloral nectaries and protection by pugnacious bodyguards. Annual Review of Ecology and Systematics 8: 407–427.
-
- Bhattacharya R, Saha S, Kostina O, Muravnik L, Mitra A.. 2020. Replacing critical point drying with a low-cost chemical drying provides comparable surface image quality of glandular trichomes from leaves of Millingtonia hortensis L. f. in scanning electron micrograph. Applied Microscopy 50: 15. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

