Single-cell transcriptomic Atlas of aging macaque ocular outflow tissues
- PMID: 38366188
- PMCID: PMC11259549
- DOI: 10.1093/procel/pwad067
Single-cell transcriptomic Atlas of aging macaque ocular outflow tissues
Abstract
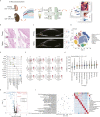
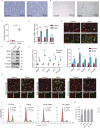
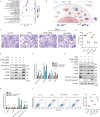
The progressive degradation in the trabecular meshwork (TM) is related to age-related ocular diseases like primary open-angle glaucoma. However, the molecular basis and biological significance of the aging process in TM have not been fully elucidated. Here, we established a dynamic single-cell transcriptomic landscape of aged macaque TM, wherein we classified the outflow tissue into 12 cell subtypes and identified mitochondrial dysfunction as a prominent feature of TM aging. Furthermore, we divided TM cells into 13 clusters and performed an in-depth analysis on cluster 0, which had the highest aging score and the most significant changes in cell proportions between the two groups. Ultimately, we found that the APOE gene was an important differentially expressed gene in cluster 0 during the aging process, highlighting the close relationship between cell migration and extracellular matrix regulation, and TM function. Our work further demonstrated that silencing the APOE gene could increase migration and reduce apoptosis by releasing the inhibition on the PI3K-AKT pathway and downregulating the expression of extracellular matrix components, thereby increasing the aqueous outflow rate and maintaining intraocular pressure within the normal range. Our work provides valuable insights for future clinical diagnosis and treatment of glaucoma.
Keywords: aging; macaque; ocular outflow tissue; single-cell transcriptomic atlas; trabecular meshwork.
© The Author(s) 2024. Published by Oxford University Press on behalf of Higher Education Press.
Conflict of interest statement
The authors declare no conflict of interests.
Figures



References
-
- Adams CM, Stacy R, Rangaswamy N. et al. Glaucoma—next generation therapeutics: impossible to possible. Pharm Res 2018;36:25. - PubMed
-
- Angello JC, Pendergrass WR, Norwood TH. et al. Proliferative potential of human fibroblasts: an inverse dependence on cell size. J Cell Physiol 2005;132:125–130. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

