A Kinetically Controlled Bioconjugation Method for the Synthesis of Radioimmunoconjugates and the Development of a Domain Mapping MS-Workflow for Its Characterization
- PMID: 38366964
- PMCID: PMC10961728
- DOI: 10.1021/acs.bioconjchem.3c00519
A Kinetically Controlled Bioconjugation Method for the Synthesis of Radioimmunoconjugates and the Development of a Domain Mapping MS-Workflow for Its Characterization
Abstract
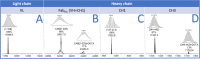
Immunoconjugates exploit the high affinity of monoclonal antibodies for a recognized antigen to selectively deliver a cytotoxic payload, such as drugs or radioactive nuclides, at the site of disease. Despite numerous techniques have been recently developed for site-selective bioconjugations of protein structures, reaction of ε-amine group of lysine residues with electrophilic reactants, such as activated esters (NHS), is the main method reported in the literature as it maintains proteins in their native conformation. Since antibodies hold a high number of lysine residues, a heterogeneous mixture of conjugates will be generated, which can result in decreased target affinity. Here, we report an intradomain regioselective bioconjugation between the monoclonal antibody Trastuzumab and the N-hydroxysuccinimide ester of the chelator 2,2',2″,2‴-(1,4,7,10-tetraazacyclododecane-1,4,7,10-tetrayl)tetraacetic acid (DOTA) by a kinetically controlled reaction adding substoichiometric quantities of the activated ester to the mAb working at slightly basic pH. Liquid chromatography-mass spectrometry (LC-MS) analyses were carried out to assess the chelator-antibody ratio (CAR) and the number of chelating moieties linked to the mAb chains. Proteolysis experiments showed four lysine residues mainly involved in bioconjugation (K188 for the light chain and K30, K293, and K417 for the heavy chain), each of which was located in a different domain. Since the displayed intradomain regioselectivity, a domain mapping MS-workflow, based on a selective domain denaturation, was developed to quantify the percentage of chelator linked to each mAb domain. The resulting immunoconjugate mixture showed an average CAR of 0.9. About a third of the heavy chains were found as monoconjugated, whereas conjugation of the chelator in the light chain was negligible. Domain mapping showed the CH3 domain bearing 13% of conjugated DOTA, followed by CH2 and VH respectively bearing 12.5 and 11% of bonded chelator. Bioconjugation was not found in the CH1 domain, whereas for the light chain, only the CL domain was conjugated (6%). Data analysis based on LC-MS quantification of different analytical levels (intact, reduced chains, and domains) provided the immunoconjugate formulation. A mixture of immunoconjugates restricted to 15 species was obtained, and the percentage of each one within the mixture was calculated. In particular, species bearing 1 DOTA with a relative abundance ranging from 4 to 20-fold, in comparison to species bearing 2DOTA, were observed. Pairing of bioconjugation under kinetic control with the developed domain mapping MS-workflow could raise the standard of chemical quality for immunoconjugates obtained with commercially available reactants.
Conflict of interest statement
The authors declare the following competing financial interest(s): The authors declare no competing financial interest. Marco A. Pometti is the inventor of application IT2024000001524 entitled “Domain Mapping”, filed on January 26, 2024.
Figures




Similar articles
-
Lysine-Directed Site-Selective Bioconjugation for the Creation of Radioimmunoconjugates.Bioconjug Chem. 2022 Sep 21;33(9):1750-1760. doi: 10.1021/acs.bioconjchem.2c00354. Epub 2022 Aug 10. Bioconjug Chem. 2022. PMID: 35946495 Free PMC article.
-
A facile, water-soluble method for modification of proteins with DOTA. Use of elevated temperature and optimized pH to achieve high specific activity and high chelate stability in radiolabeled immunoconjugates.Bioconjug Chem. 1994 Nov-Dec;5(6):565-76. doi: 10.1021/bc00030a012. Bioconjug Chem. 1994. PMID: 7873659
-
Development of a potential high-throughput workflow to characterize sites of bioconjugation by immuno-affinity capture coupled to MALDI-TOF mass spectrometry.Bioconjug Chem. 2013 Jan 16;24(1):53-62. doi: 10.1021/bc300413c. Epub 2012 Dec 12. Bioconjug Chem. 2013. PMID: 23186023
-
Practical approaches for overcoming challenges in heightened characterization of antibody-drug conjugates with new methodologies and ultrahigh-resolution mass spectrometry.MAbs. 2018 Apr;10(3):335-345. doi: 10.1080/19420862.2018.1433973. Epub 2018 Feb 20. MAbs. 2018. PMID: 29393747 Free PMC article. Review.
-
Site-selective lysine conjugation methods and applications towards antibody-drug conjugates.Chem Commun (Camb). 2021 Oct 14;57(82):10689-10702. doi: 10.1039/d1cc03976h. Chem Commun (Camb). 2021. PMID: 34570125 Free PMC article. Review.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

