This is a preprint.
Characterization, structure and inhibition of the human succinyl-CoA:glutarate-CoA transferase, a genetic modifier of glutaric aciduria type 1
- PMID: 38370847
- PMCID: PMC10871334
- DOI: 10.1101/2024.02.07.578422
Characterization, structure and inhibition of the human succinyl-CoA:glutarate-CoA transferase, a genetic modifier of glutaric aciduria type 1
Update in
-
Characterization, Structure, and Inhibition of the Human Succinyl-CoA:glutarate-CoA Transferase, a Putative Genetic Modifier of Glutaric Aciduria Type 1.ACS Chem Biol. 2024 Jul 19;19(7):1544-1553. doi: 10.1021/acschembio.4c00204. Epub 2024 Jun 24. ACS Chem Biol. 2024. PMID: 38915184 Free PMC article.
Abstract
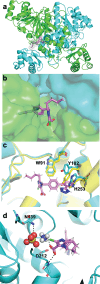
Glutaric Aciduria Type 1 (GA1) is a serious inborn error of metabolism with no pharmacological treatments. A novel strategy to treat this disease is to divert the toxic biochemical intermediates to less toxic or non-toxic metabolites. Here, we report a novel target, SUGCT, which we hypothesize suppresses the GA1 metabolic phenotype through decreasing glutaryl-CoA. We report the structure of SUGCT, the first eukaryotic structure of a type III CoA transferase, develop a high-throughput enzyme assay and a cell-based assay, and identify valsartan and losartan carboxylic acid as inhibitors of the enzyme validating the screening approach. These results may form the basis for future development of new pharmacological intervention to treat GA1.
Figures




References
-
- Deana R., Rigoni F., Deana A.D., Galzigna L., Submitochondrial localization and partial purification of the succinylCoA: 3-hydroxy-3-methylglutarate coenzyme A transferase from rat liver, Biochim Biophys Acta 662(1) (1981) 119–24. - PubMed
-
- Francesconi M.A., Donella-Deana A., Furlanetto V., Cavallini L., Palatini P., Deana R., Further purification and characterization of the succinyl-CoA:3-hydroxy-3-methylglutarate coenzyme A transferase from rat-liver mitochondria, Biochim Biophys Acta 999(2) (1989) 163–70. - PubMed
-
- Deana R., Substrate specificity of a dicarboxyl-CoA: dicarboxylic acid coenzyme A transferase from rat liver mitochondria, Biochem Int 26(4) (1992) 767–73. - PubMed
-
- Rangarajan E.S., Li Y., Iannuzzi P., Cygler M., Matte A., Crystal structure of Escherichia coli crotonobetainyl-CoA: carnitine CoA-transferase (CaiB) and its complexes with CoA and carnitinyl-CoA, Biochemistry 44(15) (2005) 5728–38. - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
