NAD+ prevents septic shock-induced death by non-canonical inflammasome blockade and IL-10 cytokine production in macrophages
- PMID: 38372712
- PMCID: PMC10942599
- DOI: 10.7554/eLife.88686
NAD+ prevents septic shock-induced death by non-canonical inflammasome blockade and IL-10 cytokine production in macrophages
Abstract
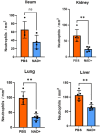
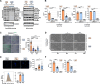
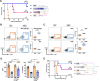
Septic shock is characterized by an excessive inflammatory response depicted in a cytokine storm that results from invasive bacterial, fungi, protozoa, and viral infections. Non-canonical inflammasome activation is crucial in the development of septic shock promoting pyroptosis and proinflammatory cytokine production via caspase-11 and gasdermin D (GSDMD). Here, we show that NAD+ treatment protected mice toward bacterial and lipopolysaccharide (LPS)-induced endotoxic shock by blocking the non-canonical inflammasome specifically. NAD+ administration impeded systemic IL-1β and IL-18 production and GSDMD-mediated pyroptosis of macrophages via the IFN-β/STAT-1 signaling machinery. More importantly, NAD+ administration not only improved casp-11 KO (knockout) survival but rendered wild type (WT) mice completely resistant to septic shock via the IL-10 signaling pathway that was independent from the non-canonical inflammasome. Here, we delineated a two-sided effect of NAD+ blocking septic shock through a specific inhibition of the non-canonical inflammasome and promoting immune homeostasis via IL-10, underscoring its unique therapeutic potential.
Keywords: E. coli; cell biology; gasdermin D; immunology; inflammasome; inflammation; interleukin 10; nicotinamide dinucleotide; septic shock.
© 2023, Iske, El Fatimy, Nian et al.
Conflict of interest statement
JI, RE, YN, AG, SE, HC, AV, AE No competing interests declared
Figures






Update of
- doi: 10.1101/2020.03.29.013649
- doi: 10.7554/eLife.88686.1
- doi: 10.7554/eLife.88686.2
Similar articles
-
The Protective Effects of Goitrin on LPS-Induced Septic Shock in C57BL/6J Mice via Caspase-11 Non-Canonical Inflammasome Inhibition.Molecules. 2023 Mar 23;28(7):2883. doi: 10.3390/molecules28072883. Molecules. 2023. PMID: 37049646 Free PMC article.
-
[Isodopharicin C inhibits NLRP3 inflammasome activation and alleviates septic shock in mice].Nan Fang Yi Ke Da Xue Xue Bao. 2023 Sep 20;43(9):1476-1484. doi: 10.12122/j.issn.1673-4254.2023.09.04. Nan Fang Yi Ke Da Xue Xue Bao. 2023. PMID: 37814861 Free PMC article. Chinese.
-
The oxidized phospholipid oxPAPC protects from septic shock by targeting the non-canonical inflammasome in macrophages.Nat Commun. 2018 Mar 8;9(1):996. doi: 10.1038/s41467-018-03409-3. Nat Commun. 2018. PMID: 29520027 Free PMC article.
-
Caspase-11 non-canonical inflammasome: a critical sensor of intracellular lipopolysaccharide in macrophage-mediated inflammatory responses.Immunology. 2017 Oct;152(2):207-217. doi: 10.1111/imm.12787. Epub 2017 Jul 31. Immunology. 2017. PMID: 28695629 Free PMC article. Review.
-
The noncanonical inflammasome-induced pyroptosis and septic shock.Semin Immunol. 2023 Nov;70:101844. doi: 10.1016/j.smim.2023.101844. Epub 2023 Sep 29. Semin Immunol. 2023. PMID: 37778179 Review.
Cited by
-
The impact of aging on neutrophil functions and the contribution to periodontitis.Int J Oral Sci. 2025 Jan 16;17(1):10. doi: 10.1038/s41368-024-00332-w. Int J Oral Sci. 2025. PMID: 39819982 Free PMC article. Review.
-
NAD(H)-loaded nanoparticles for efficient sepsis therapy via modulating immune and vascular homeostasis.Nat Nanotechnol. 2022 Aug;17(8):880-890. doi: 10.1038/s41565-022-01137-w. Epub 2022 Jun 6. Nat Nanotechnol. 2022. PMID: 35668170 Free PMC article.
-
The Modulation of Septic Shock: A Proteomic Approach.Int J Mol Sci. 2024 Oct 3;25(19):10641. doi: 10.3390/ijms251910641. Int J Mol Sci. 2024. PMID: 39408970 Free PMC article.
-
Unraveling immunosenescence in sepsis: from cellular mechanisms to therapeutics.Cell Death Dis. 2025 May 16;16(1):393. doi: 10.1038/s41419-025-07714-w. Cell Death Dis. 2025. PMID: 40379629 Free PMC article. Review.
-
Meta-analysis of niacin and NAD metabolite treatment in infectious disease animal studies suggests benefit but requires confirmation in clinically relevant models.Sci Rep. 2025 Apr 12;15(1):12621. doi: 10.1038/s41598-025-95735-y. Sci Rep. 2025. PMID: 40221506 Free PMC article.
References
-
- Begue B, Verdier J, Rieux-Laucat F, Goulet O, Morali A, Canioni D, Hugot J-P, Daussy C, Verkarre V, Pigneur B, Fischer A, Klein C, Cerf-Bensussan N, Ruemmele FM. Defective IL10 signaling defining a subgroup of patients with inflammatory bowel disease. The American Journal of Gastroenterology. 2011;106:1544–1555. doi: 10.1038/ajg.2011.112. - DOI - PubMed
-
- Berg DJ, Kühn R, Rajewsky K, Müller W, Menon S, Davidson N, Grünig G, Rennick D. Interleukin-10 is a central regulator of the response to LPS in murine models of endotoxic shock and the Shwartzman reaction but not endotoxin tolerance. The Journal of Clinical Investigation. 1995;96:2339–2347. doi: 10.1172/JCI118290. - DOI - PMC - PubMed
-
- Bullock B, Benham MD. In: StatPearls. Bullock B, editor. StatPearls Publishing StatPearls Publishing LLC; 2019. Bacterial sepsis. - PubMed
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

