Bioactive nanomaterials kickstart early repair processes and potentiate temporally modulated healing of healthy and diabetic wounds
- PMID: 38373363
- PMCID: PMC11658459
- DOI: 10.1016/j.biomaterials.2024.122496
Bioactive nanomaterials kickstart early repair processes and potentiate temporally modulated healing of healthy and diabetic wounds
Abstract
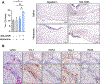
Slow-healing and chronic wounds represent a major global economic and medical burden, and there is significant unmet need for novel therapies which act to both accelerate wound closure and enhance biomechanical recovery of the skin. Here, we report a new approach in which bioactives that augment early stages of wound healing can kickstart and engender effective wound closure in healthy and diabetic, obese animals, and set the stage for subsequent tissue repair processes. We demonstrate that a nanomaterial dressing made of silk fibroin and gold nanorods (GNR) stimulates a pro-neutrophilic, innate immune, and controlled inflammatory wound transcriptomic response. Further, Silk-GNR, lasered into the wound bed, in combination with exogeneous histamine, accelerates early-stage processes in tissue repair leading to effective wound closure. Silk-GNR and histamine enhanced biomechanical recovery of skin, increased transient neoangiogenesis, myofibroblast activation, epithelial-to-mesenchymal transition (EMT) of keratinocytes and a pro-resolving neutrophilic immune response, which are hitherto unknown activities for these bioactives. Predictive and temporally coordinated delivery of growth factor nanoparticles that modulate later stages of tissue repair further accelerated wound closure in healthy and diabetic, obese animals. Our approach of kickstarting healing by delivering the "right bioactive at the right time" stimulates a multifactorial, pro-reparative response by augmenting endogenous healing and immunoregulatory mechanisms and highlights new targets to promote tissue repair.
Copyright © 2024 Elsevier Ltd. All rights reserved.
Conflict of interest statement
Declaration of competing interest The authors declare the following financial interests/personal relationships which may be considered as potential competing interests: Kaushal Rege reports financial support was provided by National Institutes of Health and Flinn Foundation. Jordan Robin Yaron reports financial support was provided by National Institutes of Health and Flinn Foundation. Kaushal Rege reports a relationship with Synergyan, LLC and Endotat Biotechnologies, LLC that includes: board membership and equity or stocks. Jordan Robin Yaron reports a relationship with Vivo Bioconsulting, LLC and Endotat Biotechnologies, LLC that includes: board membership and equity or stocks. Kaushal Rege and Deepanjan Ghosh have patent #US20210100927A1 pending to Arizona Board of Regents of ASU.
Figures
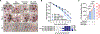




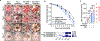

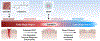
References
-
- Reinke JM, Sorg H, Wound repair and regeneration, Eur Surg Res 49(1) (2012) 35–43. - PubMed
-
- Sun BK, Siprashvili Z, Khavari PA, Advances in skin grafting and treatment of cutaneous wounds, Science 346(6212) (2014) 941–5. - PubMed
-
- Powers JG, Higham C, Broussard K, Phillips TJ, Wound healing and treating wounds: Chronic wound care and management, Journal of the American Academy of Dermatology 74(4) (2016) 607–625. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

