mGluR7 allosteric modulator AMN082 corrects protein synthesis and pathological phenotypes in FXS
- PMID: 38374465
- PMCID: PMC10940663
- DOI: 10.1038/s44321-024-00038-w
mGluR7 allosteric modulator AMN082 corrects protein synthesis and pathological phenotypes in FXS
Abstract
Fragile X syndrome (FXS) is the leading cause of inherited autism and intellectual disabilities. Aberrant protein synthesis due to the loss of fragile X messenger ribonucleoprotein (FMRP) is the major defect in FXS, leading to a plethora of cellular and behavioral abnormalities. However, no treatments are available to date. In this study, we found that activation of metabotropic glutamate receptor 7 (mGluR7) using a positive allosteric modulator named AMN082 represses protein synthesis through ERK1/2 and eIF4E signaling in an FMRP-independent manner. We further demonstrated that treatment of AMN082 leads to a reduction in neuronal excitability, which in turn ameliorates audiogenic seizure susceptibility in Fmr1 KO mice, the FXS mouse model. When evaluating the animals' behavior, we showed that treatment of AMN082 reduces repetitive behavior and improves learning and memory in Fmr1 KO mice. This study uncovers novel functions of mGluR7 and AMN082 and suggests the activation of mGluR7 as a potential therapeutic approach for treating FXS.
Keywords: Autism; FMRP; FXS; Protein Synthesis; mGluR7.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures





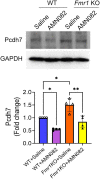
References
-
- Baker KB, Wray SP, Ritter R, Mason S, Lanthorn TH, Savelieva K., V Male and female Fmr1 knockout mice on C57 albino background exhibit spatial learning and memory impairments. Genes Brain Behav. 2010;9:562–74. - PubMed
-
- Berry-Kravis E, Des Portes V, Hagerman R, Jacquemont S, Charles P, Visootsak J, Brinkman M, Rerat K, Koumaras B, Zhu L, et al. Mavoglurant in fragile X syndrome: Results of two randomized, double-blind, placebo-controlled trials. Sci Transl Med. 2016;8:321. - PubMed
-
- Berry-Kravis E, Hicar M, Ciurlionis R. Reduced cyclic AMP production in fragile X syndrome: cytogenetic and molecular correlations. Pediatr Res. 1995;38:638–643. - PubMed
-
- Berry-Kravis E, Raspa M, Loggin-Hester L, Bishop E, Holiday D, Bailey DB. Seizures in fragile X syndrome: characteristics and comorbid diagnoses. Am J Intellect Dev Disabil. 2010;115:461–472. - PubMed
-
- Berry-Kravis E, Sklena P. Demonstration of abnormal cyclic AMP production in platelets from patients with fragile X syndrome. Am J Med Genet. 1993;45:81–87. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

