Endogenous IL-27 during toxoplasmosis limits early monocyte responses and their inflammatory activation by pathological T cells
- PMID: 38376210
- PMCID: PMC10936422
- DOI: 10.1128/mbio.00083-24
Endogenous IL-27 during toxoplasmosis limits early monocyte responses and their inflammatory activation by pathological T cells
Abstract
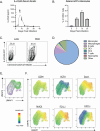
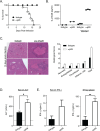
Mice that lack the genes for IL-27, or the IL-27 receptor, and infected with Toxoplasma gondii develop T cell-mediated pathology. Here, studies were performed to determine the impact of endogenous IL-27 on the immune response to T. gondii in wild-type (WT) mice. Analysis of infected mice revealed the early production of IL-27p28 by a subset of Ly6Chi, inflammatory monocytes, and sustained IL-27p28 production at sites of acute and chronic infection. Administration of anti-IL-27p28 prior to infection resulted in an early (day 5) increase in levels of macrophage and granulocyte activation, as well as enhanced effector T cell responses, as measured by both cellularity, cytokine production, and transcriptional profiling. This enhanced acute response led to immune pathology, while blockade during the chronic phase of infection resulted in enhanced T cell responses but no systemic pathology. In the absence of IL-27, the enhanced monocyte responses observed at day 10 were a secondary consequence of activated CD4+ T cells. Thus, in WT mice, IL-27 has distinct suppressive effects that impact innate and adaptive immunity during different phases of this infection.
Importance: The molecule IL-27 is critical in limiting the immune response to the parasite Toxoplasma gondii. In the absence of IL-27, a lethal, overactive immune response develops during infection. However, when exactly in the course of infection this molecule is needed was unclear. By selectively inhibiting IL-27 during this parasitic infection, we discovered that IL-27 was only needed during, but not prior to, infection. Additionally, IL-27 is only needed in the active areas in which the parasite is replicating. Finally, our work found that a previously unstudied cell type, monocytes, was regulated by IL-27, which contributes further to our understanding of the regulatory networks established by this molecule.
Keywords: IL-27p28; immunity; inflammation; neutralizing antibody; toxoplasma.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Toxoplasma gondii parasites induce a localized myeloid cell immune response surrounding parasites in the brain during acute infection.mBio. 2025 Jul 9;16(7):e0081025. doi: 10.1128/mbio.00810-25. Epub 2025 Jun 10. mBio. 2025. PMID: 40492741 Free PMC article.
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2021 Apr 19;4(4):CD011535. doi: 10.1002/14651858.CD011535.pub4. Cochrane Database Syst Rev. 2021. Update in: Cochrane Database Syst Rev. 2022 May 23;5:CD011535. doi: 10.1002/14651858.CD011535.pub5. PMID: 33871055 Free PMC article. Updated.
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2017 Dec 22;12(12):CD011535. doi: 10.1002/14651858.CD011535.pub2. Cochrane Database Syst Rev. 2017. Update in: Cochrane Database Syst Rev. 2020 Jan 9;1:CD011535. doi: 10.1002/14651858.CD011535.pub3. PMID: 29271481 Free PMC article. Updated.
-
Antiretrovirals for reducing the risk of mother-to-child transmission of HIV infection.Cochrane Database Syst Rev. 2011 Jul 6;(7):CD003510. doi: 10.1002/14651858.CD003510.pub3. Cochrane Database Syst Rev. 2011. PMID: 21735394
-
Toxoplasma gondii Dissemination in the Brain Is Facilitated by Infiltrating Peripheral Immune Cells.mBio. 2022 Dec 20;13(6):e0283822. doi: 10.1128/mbio.02838-22. Epub 2022 Nov 29. mBio. 2022. PMID: 36445695 Free PMC article.
Cited by
-
Bisphenol S Exposure and MASLD: A Mechanistic Study in Mice.Environ Health Perspect. 2025 May;133(5):57009. doi: 10.1289/EHP17057. Epub 2025 May 14. Environ Health Perspect. 2025. PMID: 40203079 Free PMC article.
-
Interleukin-27-producing cells in gram-negative neonatal sepsis display diverse phenotypes and functions in the liver.Immunohorizons. 2025 Jul 14;9(8):vlaf026. doi: 10.1093/immhor/vlaf026. Immunohorizons. 2025. PMID: 40682363 Free PMC article.
-
IL-27 limits HSPC differentiation during infection and protects from stem cell exhaustion.bioRxiv [Preprint]. 2025 Jun 24:2025.01.15.633135. doi: 10.1101/2025.01.15.633135. bioRxiv. 2025. PMID: 39868131 Free PMC article. Preprint.
-
Interleukin-27 is antiviral at the maternal-fetal interface.Res Sq [Preprint]. 2025 Jun 5:rs.3.rs-6513464. doi: 10.21203/rs.3.rs-6513464/v1. Res Sq. 2025. PMID: 40502752 Free PMC article. Preprint.
-
Neuroinflammation and Amyotrophic Lateral Sclerosis: Recent Advances in Anti-Inflammatory Cytokines as Therapeutic Strategies.Int J Mol Sci. 2025 Apr 18;26(8):3854. doi: 10.3390/ijms26083854. Int J Mol Sci. 2025. PMID: 40332510 Free PMC article. Review.
References
-
- Pflanz S, Timans JC, Cheung J, Rosales R, Kanzler H, Gilbert J, Hibbert L, Churakova T, Travis M, Vaisberg E, Blumenschein WM, Mattson JD, Wagner JL, To W, Zurawski S, McClanahan TK, Gorman DM, Bazan JF, de Waal Malefyt R, Rennick D, Kastelein RA. 2002. IL-27, a heterodimeric cytokine composed of EBI3 and p28 protein, induces proliferation of naive CD4+ T cells. Immunity 16:779–790. doi:10.1016/s1074-7613(02)00324-2 - DOI - PubMed
-
- Kilgore AM, Welsh S, Cheney EE, Chitrakar A, Blain TJ, Kedl BJ, Hunter CA, Pennock ND, Kedl RM. 2018. IL-27P28 production by XCR1+ dendritic cells and monocytes effectively predicts adjuvant-elicited CD8+ T cell responses. Immunohorizons 2:1–11. doi:10.4049/immunohorizons.1700054 - DOI - PMC - PubMed
-
- Artis D, Villarino A, Silverman M, He W, Thornton EM, Mu S, Summer S, Covey TM, Huang E, Yoshida H, Koretzky G, Goldschmidt M, Wu GD, de Sauvage F, Miller HRP, Saris CJM, Scott P, Hunter CA. 2004. The IL-27 receptor (WSX-1) is an inhibitor of innate and adaptive elements of type 2 immunity. J Immunol 173:5626–5634. doi:10.4049/jimmunol.173.9.5626 - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

