Antibacterial Marinopyrroles and Pseudilins Act as Protonophores
- PMID: 38377384
- PMCID: PMC10949930
- DOI: 10.1021/acschembio.3c00773
Antibacterial Marinopyrroles and Pseudilins Act as Protonophores
Abstract
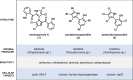
Elucidating the mechanism of action (MoA) of antibacterial natural products is crucial to evaluating their potential as novel antibiotics. Marinopyrroles, pentachloropseudilin, and pentabromopseudilin are densely halogenated, hybrid pyrrole-phenol natural products with potent activity against Gram-positive bacterial pathogens like Staphylococcus aureus. However, the exact way they exert this antibacterial activity has not been established. In this study, we explore their structure-activity relationship, determine their spatial location in bacterial cells, and investigate their MoA. We show that the natural products share a common MoA based on membrane depolarization and dissipation of the proton motive force (PMF) that is essential for cell viability. The compounds show potent protonophore activity but do not appear to destroy the integrity of the cytoplasmic membrane via the formation of larger pores or interfere with the stability of the peptidoglycan sacculus. Thus, our current model for the antibacterial MoA of marinopyrrole, pentachloropseudilin, and pentabromopseudilin stipulates that the acidic compounds insert into the membrane and transport protons inside the cell. This MoA may explain many of the deleterious biological effects in mammalian cells, plants, phytoplankton, viruses, and protozoans that have been reported for these compounds.
Conflict of interest statement
The authors declare no competing financial interest.
Figures



References
-
- Lovell F. M. The structure of a bromine-rich marine antibiotic. J. Am. Chem. Soc. 1966, 88, 4510–4511. 10.1021/ja00971a040. - DOI
-
- Cavalleri B.; Volpe G.; Tuan G.; Berti M.; Parenti F. A chlorinated phenylpyrrole antibiotic from Actinoplanes. Curr. Microbiol. An Int. J. 1978, 1, 319–324. 10.1007/BF02601691. - DOI
-
- ApSimon J. W.; Durham D. G.; Rees A. H. Synthesis of some 2-phenylpyrrole derivatives. J. Chem. Soc., Perkin Trans 1 1978, 1588–1594. 10.1039/P19780001588. - DOI
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

