Versatile dopamine-functionalized hyaluronic acid-recombinant human collagen hydrogel promoting diabetic wound healing via inflammation control and vascularization tissue regeneration
- PMID: 38379700
- PMCID: PMC10876488
- DOI: 10.1016/j.bioactmat.2024.02.010
Versatile dopamine-functionalized hyaluronic acid-recombinant human collagen hydrogel promoting diabetic wound healing via inflammation control and vascularization tissue regeneration
Abstract
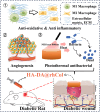
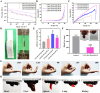
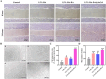
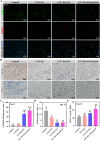
The management of chronic wounds in diabetes remains challenging due to the complexity of impaired wound healing, delayed healing, susceptibility to infection, and elevated risk of reopening, highlighting the need for effective chronic wound management with innovative approaches such as multifunctional hydrogels. Here, we have produced HA-DA@rhCol hydrogels consisting of dopamine-modified hyaluronic acid and recombinant human collagen type-III (rhCol) by oxidative coupling of the catechol group using the H2O2/HRP catalytic system. The post-reactive hydrogel has a good porous structure, swelling rate, reasonable degradation, rheological and mechanical properties, and the catechol group and dopamine impart to the hydrogel tissue adhesiveness, antioxidant capacity, and excellent photothermal effects leading to superior in vitro antimicrobial activity. In addition, the ability of rhCol to confer hydrogels to promote angiogenesis and wound repair has also been investigated. Cytotoxicity and hemolysis tests demonstrated the good biocompatibility of the hydrogel. Wound closure, collagen deposition and immunohistochemical examination confirmed the ability of the hydrogel to promote diabetic wound healing. In summary, the adhesive hemostatic antioxidative hydrogel with rhCol to promote wound healing in diabetic rat is an excellent chronic wound dressing.
Keywords: Antibacterial; Antioxidant and anti-inflammation; Diabetic wound; Hyaluronic acid hydrogel; Recombinant human collagen.
© 2024 The Authors.
Figures






References
-
- Junker J.P., Caterson E.J., Eriksson E. The microenvironment of wound healing. J. Craniofac. Surg. 2013;24:12–16. - PubMed
-
- Bandyk D.F. The diabetic foot: pathophysiology, evaluation, and treatment. Semin. Vasc. Surg. 2018;31:43–48. - PubMed
-
- Martínez García R.M., Fuentes Chacón R.M., Lorenzo Mora A.M., Ortega Anta R.M. Nutrition in the prevention and healing of chronic wounds. Importance in improving the diabetic foot. Nutr. Hosp. 2021;38:60–63. - PubMed
LinkOut - more resources
Full Text Sources

