A bispecific anti-PD-1 and PD-L1 antibody induces PD-1 cleavage and provides enhanced anti-tumor activity
- PMID: 38379869
- PMCID: PMC10877993
- DOI: 10.1080/2162402X.2024.2316945
A bispecific anti-PD-1 and PD-L1 antibody induces PD-1 cleavage and provides enhanced anti-tumor activity
Abstract
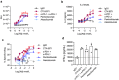
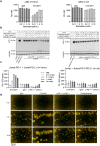
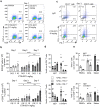
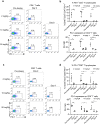
Combinatorial strategies, such as targeting different immune checkpoint receptors, hold promise to increase the breadth and duration of the response to cancer therapy. Here we describe the preclinical evaluation of CTX-8371, a protein construct which combines PD-1 and PD-L1 targeting in one bispecific, tetravalent antibody. CTX-8371 matched or surpassed the activity of anti-PD-1 and PD-L1 benchmark antibodies in several in vitro T cell activation assays and outperformed clinically approved benchmarks in the subcutaneous MC38 colon and the B16F10 lung metastasis mouse tumor models. Investigation into the mechanism of action revealed that CTX-8371 co-engagement of PD-1 and PD-L1 induced the proteolytic cleavage and loss of cell surface PD-1, which is a novel and non-redundant mechanism that adds to the PD-1/PD-L1 signaling axis blockade. The combination of CTX-8371 and an agonistic anti-CD137 antibody further increased the anti-tumor efficacy with long-lasting curative therapeutic effect. In summary, CTX-8371 is a novel checkpoint inhibitor that might provide greater clinical benefit compared to current anti-PD-1 and PD-L1 antibodies, especially when combined with agents with orthogonal mechanisms of action, such as agonistic anti-CD137 antibodies.
Keywords: Cancer immunotherapy; immuno-oncology.
© 2024 Compass Therapeutics Inc. Published with license by Taylor & Francis Group, LLC.
Conflict of interest statement
All the authors are current or former Compass Therapeutics Inc. employees and either owned or own equity in the company.
Figures




References
-
- Freeman GJ, Long AJ, Iwai Y, Bourque K, Chernova T, Nishimura H, Fitz LJ, Malenkovich N, Okazaki T, Byrne MC. et al. Engagement of the PD-1 immunoinhibitory receptor by a novel B7 family member leads to negative regulation of lymphocyte activation. J Exp Med. 2000;192(7):1027–1034. doi:10.1084/jem.192.7.1027. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
