Meiotic protein SYCP2 confers resistance to DNA-damaging agents through R-loop-mediated DNA repair
- PMID: 38383600
- PMCID: PMC10881575
- DOI: 10.1038/s41467-024-45693-2
Meiotic protein SYCP2 confers resistance to DNA-damaging agents through R-loop-mediated DNA repair
Erratum in
-
Author Correction: Meiotic protein SYCP2 confers resistance to DNA-damaging agents through R-loop-mediated DNA repair.Nat Commun. 2024 May 7;15(1):3819. doi: 10.1038/s41467-024-48241-0. Nat Commun. 2024. PMID: 38714829 Free PMC article. No abstract available.
Abstract
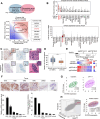
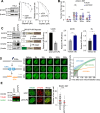
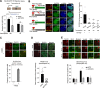
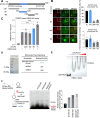
Drugs targeting the DNA damage response (DDR) are widely used in cancer therapy, but resistance to these drugs remains a major clinical challenge. Here, we show that SYCP2, a meiotic protein in the synaptonemal complex, is aberrantly and commonly expressed in breast and ovarian cancers and associated with broad resistance to DDR drugs. Mechanistically, SYCP2 enhances the repair of DNA double-strand breaks (DSBs) through transcription-coupled homologous recombination (TC-HR). SYCP2 promotes R-loop formation at DSBs and facilitates RAD51 recruitment independently of BRCA1. SYCP2 loss impairs RAD51 localization, reduces TC-HR, and renders tumors sensitive to PARP and topoisomerase I (TOP1) inhibitors. Furthermore, our studies of two clinical cohorts find that SYCP2 overexpression correlates with breast cancer resistance to antibody-conjugated TOP1 inhibitor and ovarian cancer resistance to platinum treatment. Collectively, our data suggest that SYCP2 confers cancer cell resistance to DNA-damaging agents by stimulating R-loop-mediated DSB repair, offering opportunities to improve DDR therapy.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


Similar articles
-
MePCE promotes homologous recombination through coordinating R-loop resolution at DNA double-stranded breaks.Cell Rep. 2025 Jun 24;44(6):115740. doi: 10.1016/j.celrep.2025.115740. Epub 2025 May 22. Cell Rep. 2025. PMID: 40411785 Free PMC article.
-
Suppression of homologous recombination sensitizes human tumor cells to IGF-1R inhibition.Int J Cancer. 2015 Jun 15;136(12):2961-6. doi: 10.1002/ijc.29327. Epub 2014 Nov 25. Int J Cancer. 2015. PMID: 25388513 Free PMC article.
-
Recruitment of RBM6 to DNA Double-Strand Breaks Fosters Homologous Recombination Repair.Mol Cell Biol. 2023;43(3):130-142. doi: 10.1080/10985549.2023.2187105. Mol Cell Biol. 2023. PMID: 36941773 Free PMC article.
-
BRCA1-Dependent and Independent Recruitment of PALB2-BRCA2-RAD51 in the DNA Damage Response and Cancer.Cancer Res. 2022 Sep 16;82(18):3191-3197. doi: 10.1158/0008-5472.CAN-22-1535. Cancer Res. 2022. PMID: 35819255 Free PMC article. Review.
-
Homologous recombination deficiency real-time clinical assays, ready or not?Gynecol Oncol. 2020 Dec;159(3):877-886. doi: 10.1016/j.ygyno.2020.08.035. Epub 2020 Sep 20. Gynecol Oncol. 2020. PMID: 32967790 Review.
Cited by
-
Human Papillomavirus-Encoded microRNAs as Regulators of Human Gene Expression in Anal Squamous Cell Carcinoma: A Meta-Transcriptomics Study.Noncoding RNA. 2025 Jun 9;11(3):43. doi: 10.3390/ncrna11030043. Noncoding RNA. 2025. PMID: 40559621 Free PMC article.
-
Boosting Hydroxyl Radical Generation with Nitrogen Vacancy-Modified Carbon Nitride for Triggering Dual Damage of Cancer Nucleus DNA-Mitochondria against Hypoxic Tumors.Int J Nanomedicine. 2025 Jul 5;20:8765-8781. doi: 10.2147/IJN.S515726. eCollection 2025. Int J Nanomedicine. 2025. PMID: 40635718 Free PMC article.
-
Consistently processed RNA sequencing data from 50 sources enriched for pediatric data.Sci Data. 2025 Jul 2;12(1):1134. doi: 10.1038/s41597-025-05376-z. Sci Data. 2025. PMID: 40603900 Free PMC article.
-
Single-molecule fluorescence imaging of DNA maintenance protein binding dynamics and activities on extended DNA.Curr Opin Struct Biol. 2024 Aug;87:102863. doi: 10.1016/j.sbi.2024.102863. Epub 2024 Jun 15. Curr Opin Struct Biol. 2024. PMID: 38879921 Free PMC article. Review.
-
Pathological modulation of genome maintenance by cancer/testes antigens (CTAs).DNA Repair (Amst). 2025 Mar;147:103818. doi: 10.1016/j.dnarep.2025.103818. Epub 2025 Feb 16. DNA Repair (Amst). 2025. PMID: 39983270 Review.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

