First clinical evaluation of the safety and efficacy of tarumase for the debridement of venous leg ulcers
- PMID: 38385795
- PMCID: PMC10883251
- DOI: 10.1111/iwj.14805
First clinical evaluation of the safety and efficacy of tarumase for the debridement of venous leg ulcers
Abstract
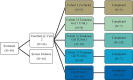
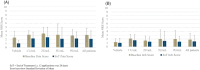
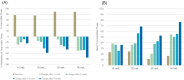
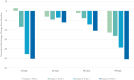
We report the first clinical evaluation of a new enzymatic wound debridement product containing tarumase in venous leg ulcer patients. As a first-in-human study, this was a prospective, open-label, multi-centre, dose escalation study across five dose cohorts and involving a total of 43 patients treated three times weekly for up to 4 weeks (12 applications). The primary and secondary endpoints of the study were to assess the systemic safety, local tolerability, and early proof of concept both for wound debridement and healing. Results indicated that the tarumase enzyme was well tolerated when applied topically to wounds, with no indications of systemic absorption, no evidence of antibody generation, and no systemic effects on coagulation pathways. Locally, there was no evidence of pain on application, no local itching, no increases in erythema, oedema, exudate or bleeding and only a few treatment emergent adverse events were reported. As the concentration of tarumase was escalated, trends towards faster and improved effectiveness of wound debridement were observed, especially in patients with significant slough at baseline. Trends towards faster rates of healing were also noted based on observations of increased granulation tissue, increased linear healing and reduction in surface area over the 4-week treatment period.
Keywords: debridement; pharmacokinetics; safety; venous leg ulcers; wound healing.
© 2024 SolasCure Ltd. International Wound Journal published by Medicalhelplines.com Inc and John Wiley & Sons Ltd.
Conflict of interest statement
David Fairlamb, Kinga Szepeshazi and David Goldsmith are employees and shareholders of SolasCure Ltd.
Figures
References
-
- Lantis J, Paedes J. Permissive maintenance debridement—the role of enzymatic debridement in chronic wound care. Wounds Int. 2017;8(2).
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

