National Rapid Genome Sequencing in Neonatal Intensive Care
- PMID: 38386321
- PMCID: PMC10884880
- DOI: 10.1001/jamanetworkopen.2024.0146
National Rapid Genome Sequencing in Neonatal Intensive Care
Abstract
Importance: National implementation of rapid trio genome sequencing (rtGS) in a clinical acute setting is essential to ensure advanced and equitable care for ill neonates.
Objective: To evaluate the feasibility, diagnostic efficacy, and clinical utility of rtGS in neonatal intensive care units (NICUs) throughout Israel.
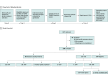
Design, setting, and participants: This prospective, public health care-based, multicenter cohort study was conducted from October 2021 to December 2022 with the Community Genetics Department of the Israeli Ministry of Health and all Israeli medical genetics institutes (n = 18) and NICUs (n = 25). Critically ill neonates suspected of having a genetic etiology were offered rtGS. All sequencing, analysis, and interpretation of data were performed in a central genomics center at Tel-Aviv Sourasky Medical Center. Rapid results were expected within 10 days. A secondary analysis report, issued within 60 days, focused mainly on cases with negative rapid results and actionable secondary findings. Pathogenic, likely pathogenic, and highly suspected variants of unknown significance (VUS) were reported.
Main outcomes and measures: Diagnostic rate, including highly suspected disease-causing VUS, and turnaround time for rapid results. Clinical utility was assessed via questionnaires circulated to treating neonatologists.
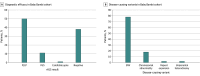
Results: A total of 130 neonates across Israel (70 [54%] male; 60 [46%] female) met inclusion criteria and were recruited. Mean (SD) age at enrollment was 12 (13) days. Mean (SD) turnaround time for rapid report was 7 (3) days. Diagnostic efficacy was 50% (65 of 130) for disease-causing variants, 11% (14 of 130) for VUS suspected to be causative, and 1 novel gene candidate (1%). Disease-causing variants included 12 chromosomal and 52 monogenic disorders as well as 1 neonate with uniparental disomy. Overall, the response rate for clinical utility questionnaires was 82% (107 of 130). Among respondents, genomic testing led to a change in medical management for 24 neonates (22%). Results led to immediate precision medicine for 6 of 65 diagnosed infants (9%), an additional 2 (3%) received palliative care, and 2 (3%) were transferred to nursing homes.
Conclusions and relevance: In this national cohort study, rtGS in critically ill neonates was feasible and diagnostically beneficial in a public health care setting. This study is a prerequisite for implementation of rtGS for ill neonates into routine care and may aid in design of similar studies in other public health care systems.
Conflict of interest statement
Figures
Comment in
-
Enhancing Neonatal Intensive Care With Rapid Genome Sequencing.JAMA Netw Open. 2024 Feb 5;7(2):e240097. doi: 10.1001/jamanetworkopen.2024.0097. JAMA Netw Open. 2024. PMID: 38386325 No abstract available.
References
-
- Murphy SL, Xu J, Kochanek KD, Arias E. Mortality in the United States, 2017. NCHS Data Brief. 2018;(328):1-8. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Miscellaneous

