Altered CD226/TIGIT expressions were associated with NK phenotypes in primary antiphospholipid syndrome and affected by IL-4/JAK pathway
- PMID: 38386917
- PMCID: PMC11036109
- DOI: 10.1093/cei/uxae016
Altered CD226/TIGIT expressions were associated with NK phenotypes in primary antiphospholipid syndrome and affected by IL-4/JAK pathway
Abstract
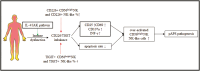
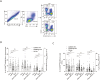
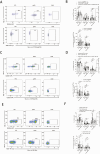
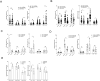
Natural killer (NK) cells were reported to be involved in the pathogenesis of primary antiphospholipid syndrome (pAPS). Immunosuppressive receptor T-cell immunoreceptor with Ig and ITIM domains (TIGIT) and activating receptor cluster of differentiation 226 (CD226) are specifically expressed on NK cells with competitive functions. This study aims to investigate the expression diversities of CD226/TIGIT on NK subsets and their associations with NK subsets activation phenotypes and potential clinical significance, furthermore, to explore potential cause for CD226/TIGIT expression diversities in pAPS. We comparatively assessed the changes of CD56brightNK, CD56dimNK, and NK-like cells in 70 pAPS patients compared with control groups, including systemic lupus erythematosus, asymptomatic antiphospholipid antibodies carriers (asymp-aPLs carriers), and healthy controls and their expression diversities of CD226/TIGIT by flow cytometry. CD25, CD69, CD107α expression, and interferon gamma (IFN-γ) secretion levels of NK subsets were detected to determine the potential association of CD226/TIGIT expression with NK subsets phenotypes. CD226/TIGIT expression levels were compared among different subgroups divided by aPLs status. Moreover, in vitro cultures were conducted to explore the potential mechanisms of CD226/TIGIT expression imbalance. CD56brightNK and CD3+CD56+NK-like cells were significantly increased while CD56dimNK cells were obviously decreased in pAPS, and CD56brightNK and NK-like cells exhibited significantly higher CD226 but lower TIGIT expressions. CD226+CD56brightNK and TIGIT-CD56brightNK cells show higher CD69 expression and IFN-γ secretion capacity, and CD226+NK-like and TIGIT-NK-like cells showed higher expressions of CD25 and CD69 but lower apoptosis rate than CD226- and TIGIT+CD56brightNK/NK-like cells, respectively. The imbalanced CD226/TIGIT expressions were most significant in aPLs triple-positive group. Imbalanced expressions of CD226/TIGIT on CD56brightNK and NK-like cells were aggravated after interleukin-4 (IL-4) stimulation and recovered after tofacitinib blocking. Our data revealed significant imbalanced CD226/TIGIT expressions on NK subsets in pAPS, which closely associated with NK subsets phenotypes and more complicated autoantibody status. CD226/TIGIT imbalanced may be affected by IL-4/Janus Kinase (JAK) pathway activation.
Keywords: CD226; IL-4; JAK; T-cell immunoreceptor with Ig and ITIM domains; natural killer cell; natural killer-like cell; primary antiphospholipid syndrome.
© The Author(s) 2024. Published by Oxford University Press on behalf of the British Society for Immunology. All rights reserved. For permissions, please e-mail: journals.permissions@oup.com.
Conflict of interest statement
None declared.
Figures



Similar articles
-
CD226+ B cells in primary Sjögren's syndrome: a key player in clinical manifestations and disease pathogenesis.Front Immunol. 2025 Jul 25;16:1623774. doi: 10.3389/fimmu.2025.1623774. eCollection 2025. Front Immunol. 2025. PMID: 40787453 Free PMC article.
-
Impact of immune checkpoint TIGIT on the activation and function of natural killer cells in rheumatoid arthritis patients.Rheumatology (Oxford). 2025 Jun 4:keaf305. doi: 10.1093/rheumatology/keaf305. Online ahead of print. Rheumatology (Oxford). 2025. PMID: 40465407
-
Expression pattern and clinical significance of MerTK on circulating NK cells in systemic lupus erythematosus.Lupus Sci Med. 2025 Jun 19;12(1):e001407. doi: 10.1136/lupus-2024-001407. Lupus Sci Med. 2025. PMID: 40541265 Free PMC article.
-
Systemic treatments for metastatic cutaneous melanoma.Cochrane Database Syst Rev. 2018 Feb 6;2(2):CD011123. doi: 10.1002/14651858.CD011123.pub2. Cochrane Database Syst Rev. 2018. PMID: 29405038 Free PMC article.
-
Adefovir dipivoxil and pegylated interferon alfa-2a for the treatment of chronic hepatitis B: a systematic review and economic evaluation.Health Technol Assess. 2006 Aug;10(28):iii-iv, xi-xiv, 1-183. doi: 10.3310/hta10280. Health Technol Assess. 2006. PMID: 16904047
Cited by
-
CD226+ B cells in primary Sjögren's syndrome: a key player in clinical manifestations and disease pathogenesis.Front Immunol. 2025 Jul 25;16:1623774. doi: 10.3389/fimmu.2025.1623774. eCollection 2025. Front Immunol. 2025. PMID: 40787453 Free PMC article.
-
The Immune Regulatory Functions of CD226 and Its Implications in Immune-Mediated Diseases.Biomolecules. 2025 Jul 14;15(7):1007. doi: 10.3390/biom15071007. Biomolecules. 2025. PMID: 40723878 Free PMC article. Review.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

