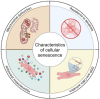
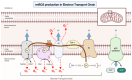
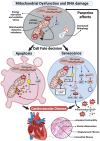
Cellular Senescence, Mitochondrial Dysfunction, and Their Link to Cardiovascular Disease
- PMID: 38391966
- PMCID: PMC10886919
- DOI: 10.3390/cells13040353
Cellular Senescence, Mitochondrial Dysfunction, and Their Link to Cardiovascular Disease
Abstract
Cardiovascular diseases (CVDs), a group of disorders affecting the heart or blood vessels, are the primary cause of death worldwide, with an immense impact on patient quality of life and disability. According to the World Health Organization, CVD takes an estimated 17.9 million lives each year, where more than four out of five CVD deaths are due to heart attacks and strokes. In the decades to come, an increased prevalence of age-related CVD, such as atherosclerosis, coronary artery stenosis, myocardial infarction (MI), valvular heart disease, and heart failure (HF) will contribute to an even greater health and economic burden as the global average life expectancy increases and consequently the world's population continues to age. Considering this, it is important to focus our research efforts on understanding the fundamental mechanisms underlying CVD. In this review, we focus on cellular senescence and mitochondrial dysfunction, which have long been established to contribute to CVD. We also assess the recent advances in targeting mitochondrial dysfunction including energy starvation and oxidative stress, mitochondria dynamics imbalance, cell apoptosis, mitophagy, and senescence with a focus on therapies that influence both and therefore perhaps represent strategies with the most clinical potential, range, and utility.
Keywords: cardiac cardiomyocyte; cardiac ischemia reperfusion; mitochondrial dysfunction; senescence.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
Similar articles
-
Mitophagy in cardiovascular disease.Clin Chim Acta. 2020 Aug;507:210-218. doi: 10.1016/j.cca.2020.04.033. Epub 2020 May 1. Clin Chim Acta. 2020. PMID: 32360616 Review.
-
Mitochondrial disorder and treatment of ischemic cardiomyopathy: Potential and advantages of Chinese herbal medicine.Biomed Pharmacother. 2023 Mar;159:114171. doi: 10.1016/j.biopha.2022.114171. Epub 2023 Jan 13. Biomed Pharmacother. 2023. PMID: 36641924 Review.
-
Endothelial mitochondrial senescence accelerates cardiovascular disease in antiretroviral-receiving HIV patients.Toxicol Lett. 2019 Dec 15;317:13-23. doi: 10.1016/j.toxlet.2019.09.018. Epub 2019 Sep 25. Toxicol Lett. 2019. PMID: 31562912 Review.
-
Role of PTEN-Induced Protein Kinase 1 as a Mitochondrial Dysfunction Regulator in Cardiovascular Disease Pathogenesis.Vasc Specialist Int. 2024 Mar 15;40:9. doi: 10.5758/vsi.230116. Vasc Specialist Int. 2024. PMID: 38486493 Free PMC article. Review.
-
Heart Disease and Ageing: The Roles of Senescence, Mitochondria, and Telomerase in Cardiovascular Disease.Subcell Biochem. 2023;103:45-78. doi: 10.1007/978-3-031-26576-1_4. Subcell Biochem. 2023. PMID: 37120464 Review.
Cited by
-
Analysis of cellular senescence-related genes in calcified aortic valve disease and the potential therapeutic role of β-Carotene.PLoS One. 2025 Mar 10;20(3):e0318574. doi: 10.1371/journal.pone.0318574. eCollection 2025. PLoS One. 2025. PMID: 40063555 Free PMC article.
-
Oxidative Stress: Signaling Pathways, Biological Functions, and Disease.MedComm (2020). 2025 Jul 1;6(7):e70268. doi: 10.1002/mco2.70268. eCollection 2025 Jul. MedComm (2020). 2025. PMID: 40599237 Free PMC article. Review.
-
The role of CXCL16 in atherosclerosis: from mechanisms to therapy.Front Immunol. 2025 May 26;16:1555438. doi: 10.3389/fimmu.2025.1555438. eCollection 2025. Front Immunol. 2025. PMID: 40491927 Free PMC article. Review.
-
Curcumin nanoparticles alleviate brain mitochondrial dysfunction and cellular senescence in γ-irradiated rats.Sci Rep. 2025 Jan 31;15(1):3857. doi: 10.1038/s41598-025-87635-y. Sci Rep. 2025. PMID: 39890961 Free PMC article.
-
Hallmarks of cellular senescence: biology, mechanisms, regulations.Exp Mol Med. 2025 Jul;57(7):1482-1491. doi: 10.1038/s12276-025-01480-7. Epub 2025 Jul 10. Exp Mol Med. 2025. PMID: 40634753 Free PMC article. Review.
References
-
- Jurk D., Wang C., Miwa S., Maddick M., Korolchuk V., Tsolou A., Gonos E.S., Thrasivoulou C., Saffrey M.J., Cameron K., et al. Postmitotic neurons develop a p21-dependent senescence-like phenotype driven by a DNA damage response. Aging Cell. 2012;11:996–1004. doi: 10.1111/j.1474-9726.2012.00870.x. - DOI - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

