Role of the osaA Gene in Aspergillus fumigatus Development, Secondary Metabolism and Virulence
- PMID: 38392775
- PMCID: PMC10890407
- DOI: 10.3390/jof10020103
Role of the osaA Gene in Aspergillus fumigatus Development, Secondary Metabolism and Virulence
Abstract
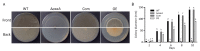
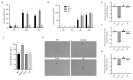
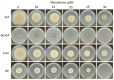
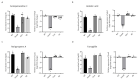
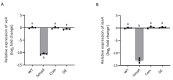
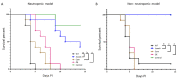
Aspergillus fumigatus is the leading cause of aspergillosis, associated with high mortality rates, particularly in immunocompromised individuals. In search of novel genetic targets against aspergillosis, we studied the WOPR transcription factor OsaA. The deletion of the osaA gene resulted in colony growth reduction. Conidiation is also influenced by osaA; both osaA deletion and overexpression resulted in a decrease in spore production. Wild-type expression levels of osaA are necessary for the expression of the conidiation regulatory genes brlA, abaA, and wetA. In addition, osaA is necessary for normal cell wall integrity. Furthermore, the deletion of osaA resulted in a reduction in the ability of A. fumigatus to adhere to surfaces, decreased thermotolerance, as well as increased sensitivity to oxidative stress. Metabolomics analysis indicated that osaA deletion or overexpression led to alterations in the production of multiple secondary metabolites, including gliotoxin. This was accompanied by changes in the expression of genes in the corresponding secondary metabolite gene clusters. These effects could be, at least in part, due to the observed reduction in the expression levels of the veA and laeA global regulators when the osaA locus was altered. Importantly, our study shows that osaA is indispensable for virulence in both neutropenic and corticosteroid-immunosuppressed mouse models.
Keywords: Aspergillus fumigatus; OsaA; WOPR domain; aspergillosis; conidiation; secondary metabolism; virulence.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures




Similar articles
-
VeA regulates conidiation, gliotoxin production, and protease activity in the opportunistic human pathogen Aspergillus fumigatus.Eukaryot Cell. 2012 Dec;11(12):1531-43. doi: 10.1128/EC.00222-12. Epub 2012 Oct 19. Eukaryot Cell. 2012. PMID: 23087369 Free PMC article.
-
The WOPR Domain Protein OsaA Orchestrates Development in Aspergillus nidulans.PLoS One. 2015 Sep 11;10(9):e0137554. doi: 10.1371/journal.pone.0137554. eCollection 2015. PLoS One. 2015. PMID: 26359867 Free PMC article.
-
The Putative APSES Transcription Factor RgdA Governs Growth, Development, Toxigenesis, and Virulence in Aspergillus fumigatus.mSphere. 2020 Nov 11;5(6):e00998-20. doi: 10.1128/mSphere.00998-20. mSphere. 2020. PMID: 33177217 Free PMC article.
-
What makes Aspergillus fumigatus a successful pathogen? Genes and molecules involved in invasive aspergillosis.Rev Iberoam Micol. 2010 Oct-Dec;27(4):155-82. doi: 10.1016/j.riam.2010.10.003. Epub 2010 Oct 23. Rev Iberoam Micol. 2010. PMID: 20974273 Review.
-
Aspergillus mycotoxins and their effect on the host.Med Mycol. 2005 May;43 Suppl 1:S95-9. doi: 10.1080/13693780500051547. Med Mycol. 2005. PMID: 16110799 Review.
Cited by
-
Recent developments in Aspergillus fumigatus research: diversity, drugs, and disease.Microbiol Mol Biol Rev. 2025 Mar 27;89(1):e0001123. doi: 10.1128/mmbr.00011-23. Epub 2025 Feb 10. Microbiol Mol Biol Rev. 2025. PMID: 39927770 Review.
-
MaPac2, a Transcriptional Regulator, Is Involved in Conidiation, Stress Tolerances and Pathogenicity in Metarhizium acridum.J Fungi (Basel). 2025 Jan 28;11(2):100. doi: 10.3390/jof11020100. J Fungi (Basel). 2025. PMID: 39997395 Free PMC article.
References
-
- Kliasova G.A., Petrova N.A., Parovichnikova E.N., Gotman L.N., Isaev V.G., Mikhaĭlova E.A., Ustinova E.N., Khoroshko N.D., Vishnevskaia E.S., Kremenetskaia A.M., et al. Invasive Pulmonary Aspergillesis. Ter. Arkh. 2005;77:65–71. - PubMed
-
- Pagano L., Girmenia C., Mele L., Ricci P., Tosti M.E., Nosari A., Buelli M., Picardi M., Allione B., Corvatta L., et al. Infections Caused by Filamentous Fungi in Patients with Hematologic Malignancies. A Report of 391 Cases by GIMEMA Infection Program. Haematologica. 2001;86:862–870. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

