Decreasing microtubule detyrosination modulates Nav1.5 subcellular distribution and restores sodium current in mdx cardiomyocytes
- PMID: 38395031
- PMCID: PMC11135645
- DOI: 10.1093/cvr/cvae043
Decreasing microtubule detyrosination modulates Nav1.5 subcellular distribution and restores sodium current in mdx cardiomyocytes
Abstract
Aims: The microtubule (MT) network plays a major role in the transport of the cardiac sodium channel Nav1.5 to the membrane, where the latter associates with interacting proteins such as dystrophin. Alterations in MT dynamics are known to impact on ion channel trafficking. Duchenne muscular dystrophy (DMD), caused by dystrophin deficiency, is associated with an increase in MT detyrosination, decreased sodium current (INa), and arrhythmias. Parthenolide (PTL), a compound that decreases MT detyrosination, has shown beneficial effects on cardiac function in DMD. We here investigated its impact on INa and Nav1.5 subcellular distribution.
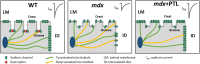
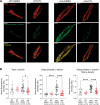
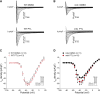
Methods and results: Ventricular cardiomyocytes (CMs) from wild-type (WT) and mdx (DMD) mice were incubated with either 10 µM PTL, 20 µM EpoY, or dimethylsulfoxide (DMSO) for 3-5 h, followed by patch-clamp analysis to assess INa and action potential (AP) characteristics in addition to immunofluorescence and stochastic optical reconstruction microscopy (STORM) to investigate MT detyrosination and Nav1.5 cluster size and density, respectively. In accordance with previous studies, we observed increased MT detyrosination, decreased INa and reduced AP upstroke velocity (Vmax) in mdx CMs compared to WT. PTL decreased MT detyrosination and significantly increased INa magnitude (without affecting INa gating properties) and AP Vmax in mdx CMs, but had no effect in WT CMs. Moreover, STORM analysis showed that in mdx CMs, Nav1.5 clusters were decreased not only in the grooves of the lateral membrane (LM; where dystrophin is localized) but also at the LM crests. PTL restored Nav1.5 clusters at the LM crests (but not at the grooves), indicating a dystrophin-independent trafficking route to this subcellular domain. Interestingly, Nav1.5 cluster density was also reduced at the intercalated disc (ID) region of mdx CMs, which was restored to WT levels by PTL. Treatment of mdx CMs with EpoY, a specific MT detyrosination inhibitor, also increased INa density, while decreasing the amount of detyrosinated MTs, confirming a direct mechanistic link.
Conclusion: Attenuating MT detyrosination in mdx CMs restored INa and enhanced Nav1.5 localization at the LM crest and ID. Hence, the reduced whole-cell INa density characteristic of mdx CMs is not only the consequence of the lack of dystrophin within the LM grooves but is also due to reduced Nav1.5 at the LM crest and ID secondary to increased baseline MT detyrosination. Overall, our findings identify MT detyrosination as a potential therapeutic target for modulating INa and subcellular Nav1.5 distribution in pathophysiological conditions.
Keywords: SCN5A; Channel trafficking; Duchenne muscular dystrophy; Ion channels; Microtubule detyrosination.
© The Author(s) 2024. Published by Oxford University Press on behalf of the European Society of Cardiology.
Conflict of interest statement
Conflict of interest: none declared.
Figures





Comment in
-
Microtubules: highway to … arrhythmia?Cardiovasc Res. 2024 May 29;120(7):671-672. doi: 10.1093/cvr/cvae072. Cardiovasc Res. 2024. PMID: 38637305 No abstract available.
References
-
- Nogales E. Structural insight into microtubule function. Annu Rev Biophys Biomol Struct 2001;30:397–420. - PubMed
-
- Schuldt M, Pei J, Harakalova M, Dorsch LM, Schlossarek S, Mokry M, Knol JC, Pham TV, Schelfhorst T, Piersma SR, Dos Remedios C, Dalinghaus M, Michels M, Asselbergs FW, Moutin MJ, Carrier L, Jimenez CR, van der Velden J, Kuster DWD. Proteomic and functional studies reveal detyrosinated tubulin as treatment target in sarcomere mutation-induced hypertrophic cardiomyopathy. Circ Heart Fail 2021;14:e007022. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- Netherlands Organisation for Health Research and Development
- Dutch Federation of University Medical Centers
- Netherlands CardioVascular Research Initiative
- 91714371/Innovational Research Incentives Scheme Vidi
- ZONMW_/ZonMw/Netherlands
- P01 CA247773/CA/NCI NIH HHS/United States
- 2015-12/Royal Netherlands Academy of Sciences
- 1R35GM134947-01/GF/NIH HHS/United States
- Dutch Heart Foundation
- 17CVD02/Fondation Leducq
- R35 GM134947/GM/NIGMS NIH HHS/United States
- R01 AI153040/AI/NIAID NIH HHS/United States
- P01 CA247773/CA/NCI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Miscellaneous

