Repeated blood-brain barrier opening with a nine-emitter implantable ultrasound device in combination with carboplatin in recurrent glioblastoma: a phase I/II clinical trial
- PMID: 38396134
- PMCID: PMC10891097
- DOI: 10.1038/s41467-024-45818-7
Repeated blood-brain barrier opening with a nine-emitter implantable ultrasound device in combination with carboplatin in recurrent glioblastoma: a phase I/II clinical trial
Abstract
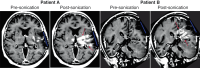
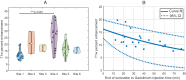
Here, the results of a phase 1/2 single-arm trial (NCT03744026) assessing the safety and efficacy of blood-brain barrier (BBB) disruption with an implantable ultrasound system in recurrent glioblastoma patients receiving carboplatin are reported. A nine-emitter ultrasound implant was placed at the end of tumor resection replacing the bone flap. After surgery, activation to disrupt the BBB was performed every four weeks either before or after carboplatin infusion. The primary objective of the Phase 1 was to evaluate the safety of escalating numbers of ultrasound emitters using a standard 3 + 3 dose escalation. The primary objective of the Phase 2 was to evaluate the efficacy of BBB opening using magnetic resonance imaging (MRI). The secondary objectives included safety and clinical efficacy. Thirty-three patients received a total of 90 monthly sonications with carboplatin administration and up to nine emitters activated without observed DLT. Grade 3 procedure-related adverse events consisted of pre syncope (n = 3), fatigue (n = 1), wound infection (n = 2), and pain at time of device connection (n = 7). BBB opening endpoint was met with 90% of emitters showing BBB disruption on MRI after sonication. In the 12 patients who received carboplatin just prior to sonication, the progression-free survival was 3.1 months, the 1-year overall survival rate was 58% and median overall survival was 14.0 months from surgery.
© 2024. The Author(s).
Conflict of interest statement
AMS and RS have received in-kind (drug) support from Bristol-Myers Squibb, in-kind (ultrasound devices) and research support from Carthera, and in-kind (drug) and research support from Agenus. AMS and RS are co-authors of intellectual property filed by Northwestern University related to therapeutic ultrasound. RS has acted or is acting as a scientific advisor or has served on advisory boards for the following companies: Alpheus Medical, AstraZeneca, Boston Scientific, Carthera, Celularity, GT Medical, Insightec, Lockwood (BlackDiamond), Northwest Biotherapeutics, Novocure, Syneos Health (Boston Biomedical), TriAct Therapeutics, and Varian Medical Systems. AMS is a consultant for Carthera and Enclear Therapeutics. MC, CD, CS, GB, and AC are employees of Carthera, inventors of patents related to the technology, or have stock ownership in Carthera. AC has received funding support from Horizon 2020 European Innovation Council; is a paid consultant of Carthera; and is part of the Board of Directors of Carthera. FD is acting as a scientific advisor or has served on advisory boards for the following companies: Novocure, Servier. AI has received research grants from Carthera, Transgene, Sanofi, Nutritheragene; travel funding from Enterome and Carthera; personal fees for advisory board from Leo Pharma, Novocure, Novartis, and Boehringer Ingelheim outside the submitted work. The remaining authors declare no competing interests.
Figures




References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

