Dysregulation of Glucocorticoid Receptor Homeostasis and Glucocorticoid-Associated Genes in Umbilical Cord Endothelial Cells of Diet-Induced Obese Pregnant Sheep
- PMID: 38396987
- PMCID: PMC10888705
- DOI: 10.3390/ijms25042311
Dysregulation of Glucocorticoid Receptor Homeostasis and Glucocorticoid-Associated Genes in Umbilical Cord Endothelial Cells of Diet-Induced Obese Pregnant Sheep
Abstract
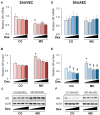
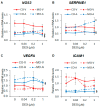
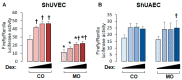
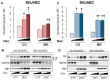
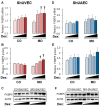
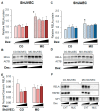
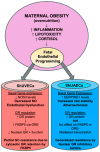
Maternal obesity (MO) is associated with offspring cardiometabolic diseases that are hypothesized to be partly mediated by glucocorticoids. Therefore, we aimed to study fetal endothelial glucocorticoid sensitivity in an ovine model of MO. Rambouillet/Columbia ewes were fed either 100% (control) or 150% (MO) National Research Council recommendations from 60 d before mating until near-term (135 days gestation). Sheep umbilical vein and artery endothelial cells (ShUVECs and ShUAECs) were used to study glucocorticoid receptor (GR) expression and function in vitro. Dexamethasone dose-response studies of gene expression, activation of a glucocorticoid response element (GRE)-dependent luciferase reporter vector, and cytosolic/nuclear GR translocation were used to assess GR homeostasis. MO significantly increased basal GR protein levels in both ShUVECs and ShUAECs. Increased GR protein levels did not result in increased dexamethasone sensitivity in the regulation of key endothelial gene expression such as endothelial nitric oxide synthase, plasminogen activator inhibitor 1, vascular endothelial growth factor, or intercellular adhesion molecule 1. In ShUVECs, MO increased GRE-dependent transactivation and FKBP prolyl isomerase 5 (FKBP5) expression. ShUAECs showed generalized glucocorticoid resistance in both dietary groups. Finally, we found that ShUVECs were less sensitive to dexamethasone-induced activation of GR than human umbilical vein endothelial cells (HUVECs). These findings suggest that MO-mediated effects in the offspring endothelium could be further mediated by dysregulation of GR homeostasis in humans as compared with sheep.
Keywords: endothelial cell; fetal programming; glucocorticoid; maternal obesity.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

Similar articles
-
Endothelial glucocorticoid receptor promoter methylation according to dexamethasone sensitivity.J Mol Endocrinol. 2015 Oct;55(2):133-46. doi: 10.1530/JME-15-0124. Epub 2015 Aug 4. J Mol Endocrinol. 2015. PMID: 26242202 Free PMC article.
-
Effect of early glucocorticoid treatment on MR and GR in late gestation ovine kidney.Kidney Int. 2002 Feb;61(2):405-13. doi: 10.1046/j.1523-1755.2002.00157.x. Kidney Int. 2002. PMID: 11849380
-
α Actinin 4 (ACTN4) Regulates Glucocorticoid Receptor-mediated Transactivation and Transrepression in Podocytes.J Biol Chem. 2017 Feb 3;292(5):1637-1647. doi: 10.1074/jbc.M116.755546. Epub 2016 Dec 20. J Biol Chem. 2017. PMID: 27998979 Free PMC article.
-
Glucocorticoids regulate mitochondrial fatty acid oxidation in fetal cardiomyocytes.J Physiol. 2021 Nov;599(21):4901-4924. doi: 10.1113/JP281860. Epub 2021 Sep 30. J Physiol. 2021. PMID: 34505639
-
How glucocorticoid receptors modulate the activity of other transcription factors: a scope beyond tethering.Mol Cell Endocrinol. 2013 Nov 5;380(1-2):41-54. doi: 10.1016/j.mce.2012.12.014. Epub 2012 Dec 23. Mol Cell Endocrinol. 2013. PMID: 23267834 Review.
Cited by
-
Perinatal glucocorticoid sensitivity in the preterm newborn: molecular mechanisms, endogenous determinants, and clinical implications.Front Endocrinol (Lausanne). 2025 Jul 16;16:1587891. doi: 10.3389/fendo.2025.1587891. eCollection 2025. Front Endocrinol (Lausanne). 2025. PMID: 40741174 Free PMC article. Review.
References
-
- Sandhu J. The Impact of Maternal Obesity on Maternal and Fetal Health. Neonatol. Today. 2021;16:10–12. doi: 10.51362/neonatology.today/202121621012. - DOI
-
- Iozzo P., Holmes M., Schmidt M.V., Cirulli F., Guzzardi M.A., Berry A., Balsevich G., Andreassi M.G., Wesselink J.-J., Liistro T., et al. Developmental ORIgins of Healthy and Unhealthy AgeiNg: The Role of Maternal Obesity-Introduction to DORIAN. Obes. Facts. 2014;7:130–151. doi: 10.1159/000362656. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

