Antioxidant Efficacy of Hwangryunhaedok-tang through Nrf2 and AMPK Signaling Pathway against Neurological Disorders In Vivo and In Vitro
- PMID: 38396988
- PMCID: PMC10889506
- DOI: 10.3390/ijms25042313
Antioxidant Efficacy of Hwangryunhaedok-tang through Nrf2 and AMPK Signaling Pathway against Neurological Disorders In Vivo and In Vitro
Abstract
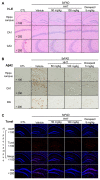
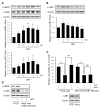
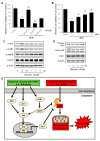
Alzheimer's disease (AD) is a representative cause of dementia and is caused by neuronal loss, leading to the accumulation of aberrant neuritic plaques and the formation of neurofibrillary tangles. Oxidative stress is involved in the impaired clearance of amyloid beta (Aβ), and Aβ-induced oxidative stress causes AD by inducing the formation of neurofibrillary tangles. Hwangryunhaedok-tang (HHT, Kracie K-09®), a traditional herbal medicine prescription, has shown therapeutic effects on various diseases. However, the studies of HHT as a potential treatment for AD are insufficient. Therefore, our study identified the neurological effects and mechanisms of HHT and its key bioactive compounds against Alzheimer's disease in vivo and in vitro. In a 5xFAD mouse model, our study confirmed that HHT attenuated cognitive impairments in the Morris water maze (MWM) test and passive avoidance (PA) test. In addition, the prevention of neuron impairment, reduction in the protein levels of Aβ, and inhibition of cell apoptosis were confirmed with brain tissue staining. In HT-22 cells, HHT attenuates tBHP-induced cytotoxicity, ROS generation, and mitochondrial dysfunction. It was verified that HHT exerts a neuroprotective effect by activating signaling pathways interacting with Nrf2, such as MAPK/ERK, PI3K/Akt, and LKB1/AMPK. Among the components, baicalein, a bioavailable compound of HHT, exhibited neuroprotective properties and activated the Akt, AMPK, and Nrf2/HO-1 pathways. Our findings indicate a mechanism for HHT and its major bioavailable compounds to treat and prevent AD and suggest its potential.
Keywords: AMPK; Alzheimer’s disease; Hwangryunhaedok-tang; Nrf2; baicalein.
Conflict of interest statement
The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures



Similar articles
-
Protective effect of sargahydroquinoic acid against Aβ25-35-evoked damage via PI3K/Akt mediated Nrf2 antioxidant defense system.Biomed Pharmacother. 2021 Dec;144:112271. doi: 10.1016/j.biopha.2021.112271. Epub 2021 Oct 4. Biomed Pharmacother. 2021. PMID: 34619494
-
Natural Dietary Supplementation of Anthocyanins via PI3K/Akt/Nrf2/HO-1 Pathways Mitigate Oxidative Stress, Neurodegeneration, and Memory Impairment in a Mouse Model of Alzheimer's Disease.Mol Neurobiol. 2018 Jul;55(7):6076-6093. doi: 10.1007/s12035-017-0798-6. Epub 2017 Nov 23. Mol Neurobiol. 2018. PMID: 29170981
-
Resveratrol Attenuates the Cytotoxicity Induced by Amyloid-β1-42 in PC12 Cells by Upregulating Heme Oxygenase-1 via the PI3K/Akt/Nrf2 Pathway.Neurochem Res. 2018 Feb;43(2):297-305. doi: 10.1007/s11064-017-2421-7. Epub 2017 Oct 31. Neurochem Res. 2018. PMID: 29090409
-
Implications of Phosphoinositide 3-Kinase-Akt (PI3K-Akt) Pathway in the Pathogenesis of Alzheimer's Disease.Mol Neurobiol. 2022 Jan;59(1):354-385. doi: 10.1007/s12035-021-02611-7. Epub 2021 Oct 26. Mol Neurobiol. 2022. PMID: 34699027 Review.
-
Oxidative stress in Alzheimer's disease: current knowledge of signaling pathways and therapeutics.Mol Biol Rep. 2024 Jan 2;51(1):48. doi: 10.1007/s11033-023-09021-z. Mol Biol Rep. 2024. PMID: 38165499 Review.
Cited by
-
Multiple Mechanisms and Therapeutic Strategies for the Involvement of AMPK in the Development of Alzheimer's Disease.Mol Neurobiol. 2025 Jul 15. doi: 10.1007/s12035-025-05162-3. Online ahead of print. Mol Neurobiol. 2025. PMID: 40665063 Review.
-
Effect of Low-Frequency, Low-Energy Pulsed Electromagnetic Fields in Neuronal and Microglial Cells Injured with Amyloid-Beta.Int J Mol Sci. 2024 Nov 29;25(23):12847. doi: 10.3390/ijms252312847. Int J Mol Sci. 2024. PMID: 39684558 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

