Selenium Compounds Affect Differently the Cytoplasmic Thiol/Disulfide State in Dermic Fibroblasts and Improve Cell Migration by Interacting with the Extracellular Matrix
- PMID: 38397757
- PMCID: PMC10886037
- DOI: 10.3390/antiox13020159
Selenium Compounds Affect Differently the Cytoplasmic Thiol/Disulfide State in Dermic Fibroblasts and Improve Cell Migration by Interacting with the Extracellular Matrix
Abstract
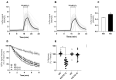
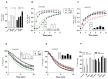
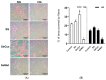
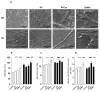
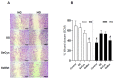
Deficient wound healing is frequently observed in patients diagnosed with diabetes, a clinical complication that compromises mobility and leads to limb amputation, decreasing patient autonomy and family lifestyle. Fibroblasts are crucial for secreting the extracellular matrix (ECM) to pave the wound site for endothelial and keratinocyte regeneration. The biosynthetic pathways involved in collagen production and crosslinking are intimately related to fibroblast redox homeostasis. In this study, two sets of human dermic fibroblasts were cultured in normal (5 mM) and high (25 mM)-glucose conditions in the presence of 1 µM selenium, as sodium selenite (inorganic) and the two selenium amino acids (organic), Se-cysteine and Se-methionine, for ten days. We investigated the ultrastructural changes in the secreted ECM induced by these conditions using scanning electron microscopy (SEM). In addition, we evaluated the redox impact of these three compounds by measuring the basal state and real-time responses of the thiol-based HyPer biosensor expressed in the cytoplasm of these fibroblasts. Our results indicate that selenium compound supplementation pushed the redox equilibrium towards a more oxidative tone in both sets of fibroblasts, and this effect was independent of the type of selenium. The kinetic analysis of biosensor responses allowed us to identify Se-cysteine as the only compound that simultaneously improved the sensitivity to oxidative stimuli and augmented the disulfide bond reduction rate in high-glucose-cultured fibroblasts. The redox response profiles showed no clear association with the ultrastructural changes observed in matrix fibers secreted by selenium-treated fibroblasts. However, we found that selenium supplementation improved the ECM secreted by high-glucose-cultured fibroblasts according to endothelial migration assessed with a wound healing assay. Direct application of sodium selenite and Se-cysteine on purified collagen fibers subjected to glycation also improved cellular migration, suggesting that these selenium compounds avoid the undesired effect of glycation.
Keywords: HyPer biosensor; cellular migration; extracellular matrix; human dermic fibroblast; selenium cysteine; selenium methionine; sodium selenite.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

Similar articles
-
Differentiated thyroid cancer and selenium supplements for protection of salivary glands from 131I treatment.Hell J Nucl Med. 2018 Jan-Apr;21(1):83-84. doi: 10.1967/s002449910712. Epub 2018 Mar 20. Hell J Nucl Med. 2018. PMID: 29550852
-
Vascular endothelial growth factor-C promotes vasculogenesis, angiogenesis, and collagen constriction in three-dimensional collagen gels.J Vasc Surg. 2005 Apr;41(4):699-707. doi: 10.1016/j.jvs.2005.01.015. J Vasc Surg. 2005. PMID: 15874936
-
Effect of selenium compounds on selenium content, growth and 35S-cystine metabolism of skin fibroblasts from normal and cystinotic individuals.Bioinorg Chem. 1976;6(3):187-202. doi: 10.1016/s0006-3061(00)80226-4. Bioinorg Chem. 1976. PMID: 1053514
-
Six-day selenium supplementation led to either UVA-photoprotection or toxic effects in human fibroblasts depending on the chemical form and dose of Se.Metallomics. 2014 Sep;6(9):1683-92. doi: 10.1039/c4mt00040d. Metallomics. 2014. PMID: 24994457
-
Selenocompounds induce a redox modulation of protein kinase C in the cell, compartmentally independent from cytosolic glutathione: its role in inhibition of tumor promotion.Arch Biochem Biophys. 1997 Dec 1;348(1):37-48. doi: 10.1006/abbi.1997.0335. Arch Biochem Biophys. 1997. PMID: 9390172
Cited by
-
Dimension reduction, cell clustering, and cell-cell communication inference for single-cell transcriptomics with DcjComm.Genome Biol. 2024 Sep 9;25(1):241. doi: 10.1186/s13059-024-03385-6. Genome Biol. 2024. PMID: 39252099 Free PMC article.
-
Semirational Design of SenC to Enhance Organic Selenium Biosynthesis.Microb Biotechnol. 2025 Mar;18(3):e70130. doi: 10.1111/1751-7915.70130. Microb Biotechnol. 2025. PMID: 40119861 Free PMC article.
References
-
- Danaei G., Finucane M.M., Lu Y., Singh G.M., Cowan M.J., Paciorek C.J., Lin J.K., Farzadfar F., Khang Y.-H., Stevens G.A., et al. National, regional, and global trends in fasting plasma glucose and diabetes prevalence since 1980: Systematic analysis of health examination surveys and epidemiological studies with 370 country-years and 2·7 million participants. Lancet. 2011;378:31–40. doi: 10.1016/S0140-6736(11)60679-X. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

