Treatment with Cobra Venom Factor Decreases Ischemic Tissue Damage in Mice
- PMID: 38397911
- PMCID: PMC10886846
- DOI: 10.3390/biomedicines12020309
Treatment with Cobra Venom Factor Decreases Ischemic Tissue Damage in Mice
Abstract
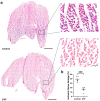
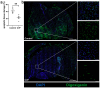
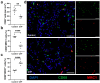
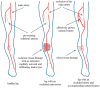
Tissue ischemia, caused by the blockage of blood vessels, can result in substantial damage and impaired tissue performance. Information regarding the functional contribution of the complement system in the context of ischemia and angiogenesis is lacking. To investigate the influence of complement activation and depletion upon femoral artery ligation (FAL), Cobra venom factor (CVF) (that functionally resembles C3b, the activated form of complement component C3) was applied in mice in comparison to control mice. Seven days after induction of muscle ischemia through FAL, gastrocnemius muscles of mice were excised and subjected to (immuno-)histological analyses. H&E and apoptotic cell staining (TUNEL) staining revealed a significant reduction in ischemic tissue damage in CVF-treated mice compared to controls. The control mice, however, exhibited a significantly higher capillary-to-muscle fiber ratio and a higher number of proliferating endothelial cells (CD31+/CD45-/BrdU+). The total number of leukocytes (CD45+) substantially decreased in CVF-treated mice versus control mice. Moreover, the CVF-treated group displayed a shift towards the M2-like anti-inflammatory and regenerative macrophage phenotype (CD68+/MRC1+). In conclusion, our findings suggest that treatment with CVF leads to reduced ischemic tissue damage along with decreased leukocyte recruitment but increased numbers of M2-like polarized macrophages, thereby enhancing tissue regeneration, repair, and healing.
Keywords: angiogenesis; cobra venom factor; complement system; inflammation; ischemia; leukocytes; macrophage polarization; macrophages; vascular occlusion.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


Similar articles
-
C3 Deficiency Leads to Increased Angiogenesis and Elevated Pro-Angiogenic Leukocyte Recruitment in Ischemic Muscle Tissue.Int J Mol Sci. 2021 May 28;22(11):5800. doi: 10.3390/ijms22115800. Int J Mol Sci. 2021. PMID: 34071589 Free PMC article.
-
Depletion of γδ T Cells Leads to Reduced Angiogenesis and Increased Infiltration of Inflammatory M1-like Macrophages in Ischemic Muscle Tissue.Cells. 2022 Apr 29;11(9):1490. doi: 10.3390/cells11091490. Cells. 2022. PMID: 35563796 Free PMC article.
-
The Absence of Extracellular Cold-Inducible RNA-Binding Protein (eCIRP) Promotes Pro-Angiogenic Microenvironmental Conditions and Angiogenesis in Muscle Tissue Ischemia.Int J Mol Sci. 2021 Aug 31;22(17):9484. doi: 10.3390/ijms22179484. Int J Mol Sci. 2021. PMID: 34502391 Free PMC article.
-
Recombinant cobra venom factor.Mol Immunol. 2004 Jun;41(2-3):191-9. doi: 10.1016/j.molimm.2004.03.011. Mol Immunol. 2004. PMID: 15159065 Review.
-
Complement depletion with humanised cobra venom factor: efficacy in preclinical models of vascular diseases.Thromb Haemost. 2015 Mar;113(3):548-52. doi: 10.1160/TH14-04-0300. Epub 2014 Jul 17. Thromb Haemost. 2015. PMID: 25031089 Review.
References
-
- Sao C.W., Aday A.W., Almarzooq Z.I., Anderson C.A., Arora P., Avery C.L., Baker-Smith C.M., Beaton A.Z., Boehme A.K., Buxton A.E., et al. Heart Disease and Stroke Statistics—2023 Update: A Report From the American Heart Association. Circulation. 2023;147:e93–e621. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

