Lipid Nanoparticle (LNP) Delivery Carrier-Assisted Targeted Controlled Release mRNA Vaccines in Tumor Immunity
- PMID: 38400169
- PMCID: PMC10891594
- DOI: 10.3390/vaccines12020186
Lipid Nanoparticle (LNP) Delivery Carrier-Assisted Targeted Controlled Release mRNA Vaccines in Tumor Immunity
Abstract
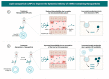
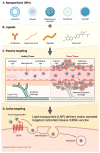
In recent years, lipid nanoparticles (LNPs) have attracted extensive attention in tumor immunotherapy. Targeting immune cells in cancer therapy has become a strategy of great research interest. mRNA vaccines are a potential choice for tumor immunotherapy, due to their ability to directly encode antigen proteins and stimulate a strong immune response. However, the mode of delivery and lack of stability of mRNA are key issues limiting its application. LNPs are an excellent mRNA delivery carrier, and their structural stability and biocompatibility make them an effective means for delivering mRNA to specific targets. This study summarizes the research progress in LNP delivery carrier-assisted targeted controlled release mRNA vaccines in tumor immunity. The role of LNPs in improving mRNA stability, immunogenicity, and targeting is discussed. This review aims to systematically summarize the latest research progress in LNP delivery carrier-assisted targeted controlled release mRNA vaccines in tumor immunity to provide new ideas and strategies for tumor immunotherapy, as well as to provide more effective treatment plans for patients.
Keywords: delivery carrier; lipid nanoparticles (LNPs); mRNA vaccine; review; tumor immunity.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures




References
-
- Bevers S., Kooijmans S.A.A., Van de Velde E., Evers M.J.W., Seghers S., Gitz-Francois J.J.J.M., van Kronenburg N.C.H., Fens M.H.A.M., Mastrobattista E., Hassler L., et al. mRNA-LNP vaccines tuned for systemic immunization induce strong antitumor immunity by engaging splenic immune cells. Mol. Ther. 2022;30:3078–3094. doi: 10.1016/j.ymthe.2022.07.007. - DOI - PMC - PubMed
-
- Fei Q., Shalosky E.M., Barnes R., Shukla V.C., Xu S., Ballinger M.N., Farkas L., Lee R.J., Ghadiali S.N., Englert J.A., et al. Macrophage-Targeted Lipid Nanoparticle Delivery of microRNA-146a to Mitigate Hemorrhagic Shock-Induced Acute Respiratory Distress Syndrome. ACS Nano. 2023;17:16539–16552. doi: 10.1021/acsnano.3c01814. - DOI - PMC - PubMed
Publication types
Grants and funding
- 81972829/National Natural Science Foundation of China
- KYQD2021096/Scientific Research Foundation of Peking University Shenzhen Hospital
- 2022ZLA006/Research and Development of Intelligent Surgical Navigation and Operating System for Precise Liver Resection
- 10001020507/Start-up Fund for Talent Researchers of Tsinghua University
- 2017ZX100203205/National Science and Technology Major Project of China
LinkOut - more resources
Full Text Sources

