Multienzymatic biotransformation of flavokawain B by entomopathogenic filamentous fungi: structural modifications and pharmacological predictions
- PMID: 38402203
- PMCID: PMC10893614
- DOI: 10.1186/s12934-024-02338-9
Multienzymatic biotransformation of flavokawain B by entomopathogenic filamentous fungi: structural modifications and pharmacological predictions
Abstract
Background: Flavokawain B is one of the naturally occurring chalcones in the kava plant (Piper methysticum). It exhibits anticancer, anti-inflammatory and antimalarial properties. Due to its therapeutic potential, flavokawain B holds promise for the treatment of many diseases. However, due to its poor bioavailability and low aqueous solubility, its application remains limited. The attachment of a sugar unit impacts the stability and solubility of flavonoids and often determines their bioavailability and bioactivity. Biotransformation is an environmentally friendly way to improve the properties of compounds, for example, to increase their hydrophilicity and thus affect their bioavailability. Recent studies proved that entomopathogenic filamentous fungi from the genera Isaria and Beauveria can perform O-methylglycosylation of hydroxyflavonoids or O-demethylation and hydroxylation of selected chalcones and flavones.
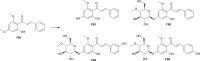
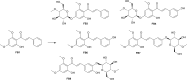
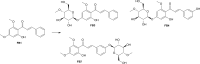
Results: In the present study, we examined the ability of entomopathogenic filamentous fungal strains of Beauveria bassiana, Beauveria caledonica, Isaria farinosa, Isaria fumosorosea, and Isaria tenuipes to transform flavokawain B into its glycosylated derivatives. The main process occurring during the reaction is O-demethylation and/or hydroxylation followed by 4-O-methylglycosylation. The substrate used was characterized by low susceptibility to transformations compared to our previously described transformations of flavones and chalcones in the cultures of the tested strains. However, in the culture of the B. bassiana KCh J1.5 and BBT, Metarhizium robertsii MU4, and I. tenuipes MU35, the expected methylglycosides were obtained with high yields. Cheminformatic analyses indicated altered physicochemical and pharmacokinetic properties in the derivatives compared to flavokawain B. Pharmacological predictions suggested potential anticarcinogenic activity, caspase 3 stimulation, and antileishmanial effects.
Conclusions: In summary, the study provided valuable insights into the enzymatic transformations of flavokawain B by entomopathogenic filamentous fungi, elucidating the structural modifications and predicting potential pharmacological activities of the obtained derivatives. The findings contribute to the understanding of the biocatalytic capabilities of these microbial cultures and the potential therapeutic applications of the modified flavokawain B derivatives.
© 2024. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
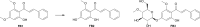
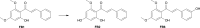
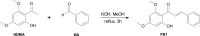
Similar articles
-
Biotransformation of Methoxyflavones by Selected Entomopathogenic Filamentous Fungi.Int J Mol Sci. 2020 Aug 25;21(17):6121. doi: 10.3390/ijms21176121. Int J Mol Sci. 2020. PMID: 32854359 Free PMC article.
-
New Glycosylated Dihydrochalcones Obtained by Biotransformation of 2'-Hydroxy-2-methylchalcone in Cultures of Entomopathogenic Filamentous Fungi.Int J Mol Sci. 2021 Sep 5;22(17):9619. doi: 10.3390/ijms22179619. Int J Mol Sci. 2021. PMID: 34502528 Free PMC article.
-
Biotransformation of Δ1-Progesterone Using Selected Entomopathogenic Filamentous Fungi and Prediction of Its Products' Bioactivity.Int J Mol Sci. 2023 Dec 29;25(1):508. doi: 10.3390/ijms25010508. Int J Mol Sci. 2023. PMID: 38203679 Free PMC article.
-
Microbial biotransformation of bioactive flavonoids.Biotechnol Adv. 2015 Jan-Feb;33(1):214-223. doi: 10.1016/j.biotechadv.2014.10.012. Epub 2014 Nov 4. Biotechnol Adv. 2015. PMID: 25447420 Review.
-
Microbial and enzymatic transformations of flavonoids.J Nat Prod. 2006 Mar;69(3):499-508. doi: 10.1021/np0504659. J Nat Prod. 2006. PMID: 16562863 Review.
Cited by
-
Antimicrobial Properties of Flavonoid Derivatives with Bromine, Chlorine, and Nitro Group Obtained by Chemical Synthesis and Biotransformation Studies.Int J Mol Sci. 2024 May 19;25(10):5540. doi: 10.3390/ijms25105540. Int J Mol Sci. 2024. PMID: 38791577 Free PMC article.
-
Insights into the quality profile and potential quality markers of Lycium barbarum (goji) berries after Tribolium castaneum infestation.Food Chem X. 2025 Jun 16;29:102668. doi: 10.1016/j.fochx.2025.102668. eCollection 2025 Jul. Food Chem X. 2025. PMID: 40599601 Free PMC article.
-
Enzymatic Glycosylation of 4'-Hydroxychalcones: Expanding the Scope of Nature's Catalytic Potential.Int J Mol Sci. 2024 Oct 25;25(21):11482. doi: 10.3390/ijms252111482. Int J Mol Sci. 2024. PMID: 39519035 Free PMC article.
-
Antimicrobial activity of new glycoside derivatives of chloroflavones obtained by fungal biotransformation.Sci Rep. 2025 Jul 16;15(1):25821. doi: 10.1038/s41598-025-11079-7. Sci Rep. 2025. PMID: 40670521 Free PMC article.
-
Bioreduction of 4'-Hydroxychalcone in Deep Eutectic Solvents: Optimization and Efficacy with Various Yeast Strains.Int J Mol Sci. 2024 Jun 28;25(13):7152. doi: 10.3390/ijms25137152. Int J Mol Sci. 2024. PMID: 39000255 Free PMC article.
References
-
- Singh YN. Kava: an overview. J Ethnopharmacol. 1992;37:13–45. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

