This is a preprint.
Mice lacking Astn2 have ASD-like behaviors and altered cerebellar circuit properties
- PMID: 38405978
- PMCID: PMC10888872
- DOI: 10.1101/2024.02.18.580354
Mice lacking Astn2 have ASD-like behaviors and altered cerebellar circuit properties
Update in
-
Mice lacking Astn2 have ASD-like behaviors and altered cerebellar circuit properties.Proc Natl Acad Sci U S A. 2024 Aug 20;121(34):e2405901121. doi: 10.1073/pnas.2405901121. Epub 2024 Aug 16. Proc Natl Acad Sci U S A. 2024. PMID: 39150780 Free PMC article.
Abstract
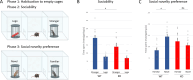
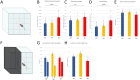
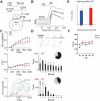
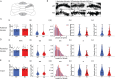
Astrotactin 2 (ASTN2) is a transmembrane neuronal protein highly expressed in the cerebellum that functions in receptor trafficking and modulates cerebellar Purkinje cell (PC) synaptic activity. We recently reported a family with a paternally inherited intragenic ASTN2 duplication with a range of neurodevelopmental disorders, including autism spectrum disorder (ASD), learning difficulties, and speech and language delay. To provide a genetic model for the role of the cerebellum in ASD-related behaviors and study the role of ASTN2 in cerebellar circuit function, we generated global and PC-specific conditional Astn2 knockout (KO and cKO, respectively) mouse lines. Astn2 KO mice exhibit strong ASD-related behavioral phenotypes, including a marked decrease in separation-induced pup ultrasonic vocalization calls, hyperactivity and repetitive behaviors, altered social behaviors, and impaired cerebellar-dependent eyeblink conditioning. Hyperactivity and repetitive behaviors were also prominent in Astn2 cKO animals. By Golgi staining, Astn2 KO PCs have region-specific changes in dendritic spine density and filopodia numbers. Proteomic analysis of Astn2 KO cerebellum reveals a marked upregulation of ASTN2 family member, ASTN1, a neuron-glial adhesion protein. Immunohistochemistry and electron microscopy demonstrates a significant increase in Bergmann glia volume in the molecular layer of Astn2 KO animals. Electrophysiological experiments indicate a reduced frequency of spontaneous excitatory postsynaptic currents (EPSCs), as well as increased amplitudes of both spontaneous EPSCs and inhibitory postsynaptic currents (IPSCs) in the Astn2 KO animals, suggesting that pre- and postsynaptic components of synaptic transmission are altered. Thus, ASTN2 regulates ASD-like behaviors and cerebellar circuit properties.
Figures




References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
