Selective Internal Radiation Therapy Using Y-90 Resin Microspheres for Metastatic Colorectal Cancer: An Updated Systematic Review and Network Meta-Analysis
- PMID: 38407790
- PMCID: PMC10960912
- DOI: 10.1007/s12325-024-02800-5
Selective Internal Radiation Therapy Using Y-90 Resin Microspheres for Metastatic Colorectal Cancer: An Updated Systematic Review and Network Meta-Analysis
Abstract
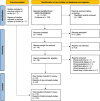
Introduction: This literature review and exploratory network meta-analysis (NMA) aimed to compare the clinical effectiveness and tolerability of selective internal radiation therapy (SIRT) using yttrium-90 (Y-90) resin microspheres, regorafenib (REG), trifluridine-tipiracil (TFD/TPI), and best supportive care (BSC) in adult patients with chemotherapy-refractory or chemotherapy-intolerant metastatic colorectal cancer (mCRC).
Methods: In light of recently published data, the literature was searched to complement and update a review published in 2018. Studies up to December 2022 comparing two or more of the treatments and reporting overall survival (OS), progression-free survival (PFS), or incidence of adverse events (AE) were included. The NMA compared hazard ratios (HRs) for OS and PFS using Markov chain Monte Carlo techniques.
Results: Fifteen studies were included, with eight studies added (none addressing SIRT). All active treatments improved OS in relation to BSC. SIRT had the longest OS among all treatments, although without statistically significant differences (HR [95% credible interval] for SIRT, 0.48 [0.27, 0.87]; TFD/TPI, 0.62 [0.46, 0.83]; REG, 0.78 [0.57, 1.05]) in a fixed effects model. Information regarding SIRT was insufficient for PFS analysis, and TFD/TPI was the best intervention (HR 2.26 [1.6, 3.18]). One SIRT study reported radioembolization-induced liver disease in > 10% of the sample; this was symptomatically managed. Non-haematological AEs (hand-foot skin reaction, fatigue, diarrhoea, hypertension, rash or desquamation) were more common with REG, while haematological events (neutropoenia, leukopenia, and anaemia) were more common with TFD/TPI.
Conclusion: Current evidence supports SIRT treatment in patients with chemotherapy-refractory or chemotherapy-intolerant mCRC compared to newer oral agents, with comparable OS and low incidence of AEs.
Keywords: Best supportive; Liver metastasis; Regorafenib; SIRT; Trifluridine–tipiracil.
© 2024. The Author(s).
Conflict of interest statement
Ion Agirrezabal was affiliated with Sirtex Medical at the time of the study. They now work for Sanofi. Victoria K. Brennan was affiliated with Sirtex Medical at the time of the study. They now work for Open Health. Phuong L. Carion was a Sirtex Medical employee at the time of the analysis. Nathalie Amoury was a Sirtex Medical employee at the time of the analysis. Suki Shergill was a Sirtex Medical employee at the time of the analysis. Bruna S. Zanotto was working on behalf of Sirtex Medical on the project. Bruna M. Vetromilla was working on behalf of Sirtex Medical on the project. André L. F. Azeredo-da-Silva was working on behalf of Sirtex Medical on the project. Patricia K. Ziegelmann was working on behalf of Sirtex Medical on the project. Victor Hugo F. de Jesus has received honoraria for academic purposes and consulting for Sirtex.
Figures
Similar articles
-
Systematic review and network meta-analyses of third-line treatments for metastatic colorectal cancer.J Cancer Res Clin Oncol. 2020 Oct;146(10):2575-2587. doi: 10.1007/s00432-020-03315-6. Epub 2020 Jul 27. J Cancer Res Clin Oncol. 2020. PMID: 32715436 Free PMC article.
-
Real-world outcomes of selective internal radiation therapy with 90Y resin microspheres in HCC/mCRC: RESIN registry Asia.Future Oncol. 2025 Aug;21(18):2287-2297. doi: 10.1080/14796694.2025.2524862. Epub 2025 Jul 15. Future Oncol. 2025. PMID: 40662675
-
A Systematic Review and Network Meta-Analysis of Regorafenib and TAS-102 in Refractory Metastatic Colorectal Cancer.Oncologist. 2019 Sep;24(9):1174-1179. doi: 10.1634/theoncologist.2019-0189. Epub 2019 Jun 4. Oncologist. 2019. PMID: 31164455 Free PMC article.
-
Effectiveness and Safety of Regorafenib and TAS-102 in Patients with Metastatic Colorectal Cancer: A Nationwide Population-Based Study in Taiwan.Cancer Res Treat. 2025 Jul;57(3):830-839. doi: 10.4143/crt.2024.376. Epub 2024 Nov 18. Cancer Res Treat. 2025. PMID: 39563201 Free PMC article.
-
Treatment options for progression or recurrence of glioblastoma: a network meta-analysis.Cochrane Database Syst Rev. 2021 May 4;5(1):CD013579. doi: 10.1002/14651858.CD013579.pub2. Cochrane Database Syst Rev. 2021. PMID: 34559423 Free PMC article.
Cited by
-
Pathogenetic development, diagnosis and clinical therapeutic approaches for liver metastasis from colorectal cancer (Review).Int J Oncol. 2025 Mar;66(3):22. doi: 10.3892/ijo.2025.5728. Epub 2025 Feb 14. Int J Oncol. 2025. PMID: 39950314 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

