Treatment guidelines for rare, early-onset conditions associated with epileptic seizures: a literature review on Rett syndrome and tuberous sclerosis complex
- PMID: 38409029
- PMCID: PMC10895812
- DOI: 10.1186/s13023-023-02994-x
Treatment guidelines for rare, early-onset conditions associated with epileptic seizures: a literature review on Rett syndrome and tuberous sclerosis complex
Abstract
Background: Rett syndrome (RTT) and tuberous sclerosis complex (TSC) are two rare disorders presenting with a range of different epileptic seizures. Seizure management requires careful therapy selection, thereby necessitating development of high-quality treatment guidelines. This targeted literature review (TLR) aimed to characterise country-specific and international treatment guidelines available for pharmacological management of seizures in RTT and TSC.
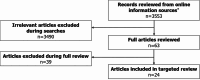
Methods: A TLR was performed between 25-Jan and 11-Mar 2021. Manual searches of online rare disease and guideline databases, and websites of national heath technology assessment bodies were conducted for the following countries: Australia, Canada, France, Germany, Israel, Italy, Japan, Spain, Switzerland, UK, and US as defined by pre-specified eligibility criteria. Search terms were developed for each condition and translated into local languages where appropriate. Eligible publications were defined as guidelines/guidance reporting pharmacological management of seizures in patients with RTT and TSC. Guideline development methodology, geographical focus, author information and treatment recommendations were extracted from guidelines. An author map was generated using R version 3.5.1 to visualise extent of collaboration between authors.
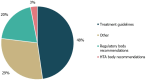
Results: 24 total guidelines were included, of which three and six contained only recommendations for RTT and TSC, respectively (some provided recommendations for ≥ 1 condition). Guideline development processes were poorly described (50% [12 guidelines] had unclear/absent literature review methodologies); reported methodologies were variable, including systematic literature reviews (SLRs)/TLRs and varying levels of expert consultation. Most (83% [20/24]) were country-specific, with guideline authors predominantly publishing in contained national groups; four guidelines were classified as 'International,' linking author groups in the US, UK, Italy and France. High levels of heterogeneity were observed in the availability of treatment recommendations across indications, with 13 and 67 recommendations found for RTT and TSC, respectively. For RTT, all treatment recommendations were positive and sodium valproate had the highest number of positive recommendations (Khwaja, Sahin (2011) Curr Opin Pediatr 23(6):633-9). All TSC treatments (21 medications) received either exclusively negative (National Organization for Rare Disorders (2019)) or positive (Chu-Shore et al. (2010) Epilepsia 51(7):1236-41) recommendations; vigabatrin received the highest number of positive recommendations (Kaur, Christodoulou (2019)).
Conclusions: This review highlights the need for the development of international high-quality and comprehensive consensus-based guidance for the management of seizures with pharmacological therapy in RTT and TSC.
Trial registration: Not applicable.
Keywords: Guidelines; Literature review; Rare diseases; Rett syndrome; Seizures; Treatment; Tuberous sclerosis complex.
© 2024. The Author(s).
Conflict of interest statement
This study was funded by GW Global Services (International) Ltd. Editorial and medical writing services were provided by Costello Medical. S. Amin, has provided consultancy and speaker services, and has participated in events and studies, for GW Global Services (International) Ltd; Medtronic Limited, Boston Scientific, Novartis, PTC therapeutics and Desitin; I. Newell, employee of Costello Medical; B. Ruban-Fell, employee of Costello Medical; J. Evans, employee of Costello Medical; K. Vyas, employee of GW Global Services (International) Ltd; C. Nortvedt, employee of GW Global Services (International) Ltd; R.F.M. Chin, has provided consultancy and speaker services, and has participated in events and studies, for GW Global Services (International) Ltd, Eisai, Zogenix, Neopharm Group and The Rize Medical Cannabis and Life Sciences UCITS ETF.
Figures






Similar articles
-
Treatment Guidelines for Rare, Early-Onset, Treatment-Resistant Epileptic Conditions: A Literature Review on Dravet Syndrome, Lennox-Gastaut Syndrome and CDKL5 Deficiency Disorder.Front Neurol. 2021 Oct 25;12:734612. doi: 10.3389/fneur.2021.734612. eCollection 2021. Front Neurol. 2021. PMID: 34759881 Free PMC article.
-
Nordic treatment guidelines for rare epileptic conditions: A literature review.Brain Behav. 2022 Jul;12(7):e2622. doi: 10.1002/brb3.2622. Epub 2022 Jun 28. Brain Behav. 2022. PMID: 35765698 Free PMC article. Review.
-
Prescription patterns of antiseizure drugs in tuberous sclerosis complex (TSC)-associated epilepsy: a multicenter cohort study from Germany and review of the literature.Expert Rev Clin Pharmacol. 2021 Jun;14(6):749-760. doi: 10.1080/17512433.2021.1911643. Epub 2021 May 31. Expert Rev Clin Pharmacol. 2021. PMID: 33792454 Review.
-
The future of Cochrane Neonatal.Early Hum Dev. 2020 Nov;150:105191. doi: 10.1016/j.earlhumdev.2020.105191. Epub 2020 Sep 12. Early Hum Dev. 2020. PMID: 33036834
-
Management of epilepsy associated with tuberous sclerosis complex: Updated clinical recommendations.Eur J Paediatr Neurol. 2018 Sep;22(5):738-748. doi: 10.1016/j.ejpn.2018.05.006. Epub 2018 May 24. Eur J Paediatr Neurol. 2018. PMID: 29880258 Review.
Cited by
-
Rett syndrome: advances in Understanding MeCP2 function, potential gene therapies, and public health implications.Mol Biol Rep. 2025 Jul 8;52(1):687. doi: 10.1007/s11033-025-10802-x. Mol Biol Rep. 2025. PMID: 40627220 Review.
References
-
- National Organization for Rare Disorders. Rett Syndrome [Internet]. 2015 [cited March 19 2020]. Available from: https://rarediseases.org/rare-diseases/rett-syndrome/.
-
- National Organization for Rare Disorders. Tuberous Sclerosis [Internet]. 2019 [cited March 19 2020]. Available from: https://rarediseases.org/rare-diseases/tuberous-sclerosis/.
-
- Merwick A, O’Brien M, Delanty N. Complex single gene disorders and Epilepsy. Epilepsia. 2012;53(Suppl 4):81–91. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

