Roles and inhibitors of FAK in cancer: current advances and future directions
- PMID: 38410129
- PMCID: PMC10895298
- DOI: 10.3389/fphar.2024.1274209
Roles and inhibitors of FAK in cancer: current advances and future directions
Abstract
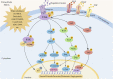
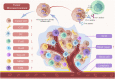
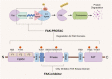
Focal adhesion kinase (FAK) is a non-receptor tyrosine kinase that exhibits high expression in various tumors and is associated with a poor prognosis. FAK activation promotes tumor growth, invasion, metastasis, and angiogenesis via both kinase-dependent and kinase-independent pathways. Moreover, FAK is crucial for sustaining the tumor microenvironment. The inhibition of FAK impedes tumorigenesis, metastasis, and drug resistance in cancer. Therefore, developing targeted inhibitors against FAK presents a promising therapeutic strategy. To date, numerous FAK inhibitors, including IN10018, defactinib, GSK2256098, conteltinib, and APG-2449, have been developed, which have demonstrated positive anti-tumor effects in preclinical studies and are undergoing clinical trials for several types of tumors. Moreover, many novel FAK inhibitors are currently in preclinical studies to advance targeted therapy for tumors with aberrantly activated FAK. The benefits of FAK degraders, especially in terms of their scaffold function, are increasingly evident, holding promising potential for future clinical exploration and breakthroughs. This review aims to clarify FAK's role in cancer, offering a comprehensive overview of the current status and future prospects of FAK-targeted therapy and combination approaches. The goal is to provide valuable insights for advancing anti-cancer treatment strategies.
Keywords: IN10018; defactinib; drug resistance; focal adhesion kinase; immune microenvironment; inhibitor; signal pathway.
Copyright © 2024 Hu, Wang, Shang, Lv, Chen, Gao and Chen.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
-
- Abhinand C. S., Galipon J., Mori M., Ramesh P., Prasad T., Raju R., et al. (2023). Temporal phosphoproteomic analysis of VEGF-A signaling in HUVECs: an insight into early signaling events associated with angiogenesis. J. Cell Commun. Signal. 17 (3), 1067–1079. 10.1007/s12079-023-00736-z - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous

