Circadian Rhythm Regulation by Pacemaker Neuron Chloride Oscillation in Flies
- PMID: 38411570
- PMCID: PMC11368518
- DOI: 10.1152/physiol.00006.2024
Circadian Rhythm Regulation by Pacemaker Neuron Chloride Oscillation in Flies
Abstract
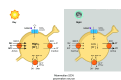
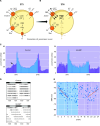
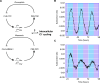
Circadian rhythms in physiology and behavior sync organisms to external environmental cycles. Here, circadian oscillation in intracellular chloride in central pacemaker neurons of the fly, Drosophila melanogaster, is reviewed. Intracellular chloride links SLC12 cation-coupled chloride transporter function with kinase signaling and the regulation of inwardly rectifying potassium channels.
Keywords: SLC12; SPAK; WNK; circadian; potassium channel.
Conflict of interest statement
No conflicts of interest, financial or otherwise, are declared by the author.
Figures
Similar articles
-
Chloride oscillation in pacemaker neurons regulates circadian rhythms through a chloride-sensing WNK kinase signaling cascade.Curr Biol. 2022 Mar 28;32(6):1429-1438.e6. doi: 10.1016/j.cub.2022.03.017. Epub 2022 Mar 17. Curr Biol. 2022. PMID: 35303418 Free PMC article.
-
Electrical silencing of PDF neurons advances the phase of non-PDF clock neurons in Drosophila.J Biol Rhythms. 2008 Apr;23(2):117-28. doi: 10.1177/0748730407312984. J Biol Rhythms. 2008. PMID: 18375861
-
Drosophila TRPA1 functions in temperature control of circadian rhythm in pacemaker neurons.J Neurosci. 2013 Apr 17;33(16):6716-25. doi: 10.1523/JNEUROSCI.4237-12.2013. J Neurosci. 2013. PMID: 23595730 Free PMC article.
-
Neurobiology of the fruit fly's circadian clock.Genes Brain Behav. 2005 Mar;4(2):65-76. doi: 10.1111/j.1601-183X.2004.00092.x. Genes Brain Behav. 2005. PMID: 15720403 Review.
-
Circadian Rhythms and Sleep in Drosophila melanogaster.Genetics. 2017 Apr;205(4):1373-1397. doi: 10.1534/genetics.115.185157. Genetics. 2017. PMID: 28360128 Free PMC article. Review.
Cited by
-
With No Lysine (K) Kinases and Sodium Transporter Function in Solute Exchange with Implications for BP Regulation as Elucidated through Drosophila.Kidney360. 2024 Oct 1;5(10):1553-1562. doi: 10.34067/KID.0000000000000564. Epub 2024 Aug 26. Kidney360. 2024. PMID: 39186374 Free PMC article. Review.
-
Function and regulation of the insect NaCCC2 sodium transport proteins.Comp Biochem Physiol A Mol Integr Physiol. 2024 Oct;296:111685. doi: 10.1016/j.cbpa.2024.111685. Epub 2024 Jun 22. Comp Biochem Physiol A Mol Integr Physiol. 2024. PMID: 38914258 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

