The Bio-Hermes Study: Biomarker database developed to investigate blood-based and digital biomarkers in community-based, diverse populations clinically screened for Alzheimer's disease
- PMID: 38415908
- PMCID: PMC11032569
- DOI: 10.1002/alz.13722
The Bio-Hermes Study: Biomarker database developed to investigate blood-based and digital biomarkers in community-based, diverse populations clinically screened for Alzheimer's disease
Abstract
Introduction: Alzheimer's disease (AD) trial participants are often screened for eligibility by brain amyloid positron emission tomography/cerebrospinal fluid (PET/CSF), which is inefficient as many are not amyloid positive. Use of blood-based biomarkers may reduce screen failures.
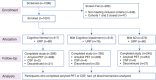
Methods: We recruited 755 non-Hispanic White, 115 Hispanic, 112 non-Hispanic Black, and 19 other minority participants across groups of cognitively normal (n = 417), mild cognitive impairment (n = 312), or mild AD (n = 272) participants. Plasma amyloid beta (Aβ)40, Aβ42, Aβ42/Aβ40, total tau, phosphorylated tau (p-tau)181, and p-tau217 were measured; amyloid PET/CSF (n = 956) determined amyloid positivity. Clinical, blood biomarker, and ethnicity/race differences associated with amyloid status were evaluated.
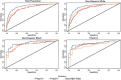
Results: Greater impairment, older age, and carrying an apolipoprotein E (apoE) ε4 allele were associated with greater amyloid burden. Areas under the receiver operating characteristic curve for amyloid status of plasma Aβ42/Aβ40, p-tau181, and p-tau217 with amyloid positivity were ≥ 0.7117 for all ethnoracial groups (p-tau217, ≥0.8128). Age and apoE ε4 adjustments and imputation of biomarker values outside limit of quantitation provided small improvement in predictive power.
Discussion: Blood-based biomarkers are highly associated with amyloid PET/CSF results in diverse populations enrolled at clinical trial sites.
Highlights: Amyloid beta (Aβ)42/Aβ40, phosphorylated tau (p-tau)181, and p-tau 217 blood-based biomarkers predicted brain amyloid positivity. P-tau 217 was the strongest predictor of brain amyloid positivity. Biomarkers from diverse ethnic, racial, and clinical cohorts predicted brain amyloid positivity. Community-based populations have similar Alzheimer's disease (AD) biomarker levels as other populations. A prescreen process with blood-based assays may reduce the number of AD trial screen failures.
Keywords: Alzheimer's disease; Alzheimer's disease blood‐based biomarkers; Alzheimer's disease ethnic and racial differences; Bio‐Hermes Study; amyloid beta 40; amyloid beta 42; amyloid beta 42/40; clinical trials; phosphorylated tau181; phosphorylated tau217; screening.
© 2024 The Authors. Alzheimer's & Dementia published by Wiley Periodicals LLC on behalf of Alzheimer's Association.
Conflict of interest statement
The following reviewed and edited the manuscript for accuracy: Richard C. Mohs, Douglas Beauregard, John Dwyer, Jennifer Gaudioso, Jason Bork, Tamiko MaGee Rodgers, Mickeal Key, Lynn Hughes, and Cyndy Cordell are employees/advisors of the Global Alzheimer's Platform Foundation. Lynn Hughes is also an advisor to IXICO PLC. Diana R. Kerwin is Founder and President of Kerwin Medical Center, Dallas, TX. Jessie Nicodemus‐Johnson and Craig Mallinckrodt are employees of Pentara Corporation. Robin Wolz is an employee of IXICO PLC. Kevin Yarasheski, Joel B. Braunstein, Tim West, Philip Verghese, Kris Kirmess, and Matthew Meyer are employees of C2N Diagnostics. David Wilson is an employee of Quanterix, and experts employed by Eli Lilly and Company. Author disclosures are available in the supporting information.
Figures
Similar articles
-
Plasma phosphorylated tau 217 and phosphorylated tau 181 as biomarkers in Alzheimer's disease and frontotemporal lobar degeneration: a retrospective diagnostic performance study.Lancet Neurol. 2021 Sep;20(9):739-752. doi: 10.1016/S1474-4422(21)00214-3. Lancet Neurol. 2021. PMID: 34418401 Free PMC article.
-
Plasma p-tau217 and p-tau217/Aβ1-42 are effective biomarkers for identifying CSF- and PET imaging-diagnosed Alzheimer's disease: Insights for research and clinical practice.Alzheimers Dement. 2025 Feb;21(2):e14536. doi: 10.1002/alz.14536. Epub 2025 Jan 30. Alzheimers Dement. 2025. PMID: 39887504 Free PMC article.
-
Combining plasma Aβ and p-tau217 improves detection of brain amyloid in non-demented elderly.Alzheimers Res Ther. 2024 May 23;16(1):115. doi: 10.1186/s13195-024-01469-w. Alzheimers Res Ther. 2024. PMID: 38778353 Free PMC article.
-
Predictive Accuracy of Blood-Derived Biomarkers for Amyloid-β Brain Deposition Along with the Alzheimer's Disease Continuum: A Systematic Review.J Alzheimers Dis. 2021;84(1):393-407. doi: 10.3233/JAD-210496. J Alzheimers Dis. 2021. PMID: 34542072
-
Plasma p-tau immunoassays in clinical research for Alzheimer's disease.Alzheimers Dement. 2025 Jan;21(1):e14397. doi: 10.1002/alz.14397. Epub 2024 Dec 3. Alzheimers Dement. 2025. PMID: 39625101 Free PMC article. Review.
Cited by
-
Cognivue Clarity characterizes mild cognitive impairment and Alzheimer's disease in biomarker confirmed cohorts in the Bio-Hermes Study.Sci Rep. 2024 Oct 18;14(1):24519. doi: 10.1038/s41598-024-75304-5. Sci Rep. 2024. PMID: 39424626 Free PMC article.
-
Diagnostic and discriminative accuracy of plasma phosphorylated tau 217 for symptomatic Alzheimer's disease in a Chinese cohort.J Prev Alzheimers Dis. 2025 May;12(5):100092. doi: 10.1016/j.tjpad.2025.100092. Epub 2025 Feb 12. J Prev Alzheimers Dis. 2025. PMID: 39948000 Free PMC article.
-
Alzheimer's disease biomarkers and their current use in clinical research and practice.Mol Psychiatry. 2025 Jan;30(1):272-284. doi: 10.1038/s41380-024-02709-z. Epub 2024 Sep 4. Mol Psychiatry. 2025. PMID: 39232196 Review.
-
Plasma p-tau217 concordance with amyloid PET among ethnically diverse older adults.Alzheimers Dement (Amst). 2024 Jul 17;16(3):e12617. doi: 10.1002/dad2.12617. eCollection 2024 Jul-Sep. Alzheimers Dement (Amst). 2024. PMID: 39021585 Free PMC article.
-
Influence of cognitive impairment and race on plasma p-Tau217 in two diverse cohorts.Alzheimers Dement. 2025 Feb;21(2):e14585. doi: 10.1002/alz.14585. Alzheimers Dement. 2025. PMID: 39988568 Free PMC article.
References
-
- Malzbender K, Lavin‐Mena L, Hughes L, Bose N, Goldman D, Patel D. Key Barriers to Clinical Trials for Alzheimer's Disease. University of Southern California Schaeffer, Leonard D Schaeffer Center for Health Policy & Economics. August 2020. Accessed March 18, 2023. https://healthpolicy.usc.edu/wp‐content/uploads/2020/08/Key‐Barriers‐to‐...
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

