Expression of GPX4 by oncolytic vaccinia virus can significantly enhance CD8+T cell function and its impact against pancreatic ductal adenocarcinoma
- PMID: 38419758
- PMCID: PMC10900272
- DOI: 10.1080/2162402X.2024.2322173
Expression of GPX4 by oncolytic vaccinia virus can significantly enhance CD8+T cell function and its impact against pancreatic ductal adenocarcinoma
Abstract
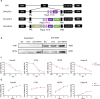
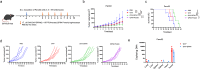
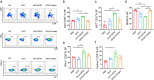
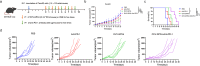
Pancreatic ductal adenocarcinoma (PDAC) is currently difficult to treat, even when therapies are combined with immune checkpoint blockade (ICB). A novel strategy for immunotherapy would be to maximize the therapeutic potential of oncolytic viruses (OVs), which have been proven to engage the regulation of tumor microenvironment (TME) and cause-specific T-cell responses. To boost tumor sensitivity to ICB therapy, this study aimed to investigate how glutathione peroxide 4 (GPX4)-loaded OVs affect CD8+ T cells and repair the immunosuppressive environment. Here, we successfully constructed a novel recombinant oncolytic vaccinia virus (OVV) encoding the mouse GPX4 gene. We found the OVV-GPX4 effectively replicated in tumor cells and prompted the expression of GPX4 in T cells. Our research indicated that OVV-GPX4 could reshape the TME, rectify the depletion of CD8+T cells, and enhance the antitumor effects of ICB therapy.
Keywords: Glutathione peroxide 4; oncolytic vaccinia virus; pancreatic ductal adenocarcinoma; tumor microenvironment.
© 2024 The Author(s). Published with license by Taylor & Francis Group, LLC.
Conflict of interest statement
No potential conflict of interest was reported by the author(s).
Figures




Similar articles
-
An oncolytic vaccinia virus encoding hyaluronidase reshapes the extracellular matrix to enhance cancer chemotherapy and immunotherapy.J Immunother Cancer. 2024 Mar 7;12(3):e008431. doi: 10.1136/jitc-2023-008431. J Immunother Cancer. 2024. PMID: 38458640 Free PMC article.
-
CD40L-armed oncolytic herpes simplex virus suppresses pancreatic ductal adenocarcinoma by facilitating the tumor microenvironment favorable to cytotoxic T cell response in the syngeneic mouse model.J Immunother Cancer. 2022 Jan;10(1):e003809. doi: 10.1136/jitc-2021-003809. J Immunother Cancer. 2022. PMID: 35086948 Free PMC article.
-
Enhanced antitumor efficacy of a novel oncolytic vaccinia virus encoding a fully monoclonal antibody against T-cell immunoglobulin and ITIM domain (TIGIT).EBioMedicine. 2021 Feb;64:103240. doi: 10.1016/j.ebiom.2021.103240. Epub 2021 Feb 10. EBioMedicine. 2021. PMID: 33581644 Free PMC article.
-
Oncolytic virotherapy for pancreatic ductal adenocarcinoma: A glimmer of hope after years of disappointment?Cytokine Growth Factor Rev. 2020 Dec;56:141-148. doi: 10.1016/j.cytogfr.2020.07.015. Epub 2020 Aug 8. Cytokine Growth Factor Rev. 2020. PMID: 32859494 Review.
-
Is oncolytic adenoviral-mediated immunotherapy through p53-overexpression the solution to refractory pancreatic ductal adenocarcinoma?Expert Rev Gastroenterol Hepatol. 2024 Jun;18(6):223-226. doi: 10.1080/17474124.2024.2363222. Epub 2024 Jun 17. Expert Rev Gastroenterol Hepatol. 2024. PMID: 38818792 Review.
Cited by
-
Ferroptosis of T cell in inflammation and tumour immunity.Clin Transl Med. 2025 Mar;15(3):e70253. doi: 10.1002/ctm2.70253. Clin Transl Med. 2025. PMID: 40045458 Free PMC article. Review.
-
GM-CSF and IL-21-armed oncolytic vaccinia virus significantly enhances anti-tumor activity and synergizes with anti-PD1 immunotherapy in pancreatic cancer.Front Immunol. 2025 Jan 3;15:1506632. doi: 10.3389/fimmu.2024.1506632. eCollection 2024. Front Immunol. 2025. PMID: 39830516 Free PMC article.
-
Fucoxanthin Induces Ferroptosis in Cancer Cells via Downregulation of the Nrf2/HO-1/GPX4 Pathway.Molecules. 2024 Jun 14;29(12):2832. doi: 10.3390/molecules29122832. Molecules. 2024. PMID: 38930897 Free PMC article.
References
-
- Farren MR, Sayegh L, Ware MB, Chen H-R, Gong J, Liang Y, Krasinskas A, Maithel SK, Zaidi M, Sarmiento JM, et al. Immunologic alterations in the pancreatic cancer microenvironment of patients treated with neoadjuvant chemotherapy and radiotherapy. JCI Insight. 2020;5(1):Insight 5: 2020. doi: 10.1172/jci.insight.130362. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
