Trypanosoma cruzi interaction with host tissues modulate the composition of large extracellular vesicles
- PMID: 38424216
- PMCID: PMC10904747
- DOI: 10.1038/s41598-024-55302-3
Trypanosoma cruzi interaction with host tissues modulate the composition of large extracellular vesicles
Abstract
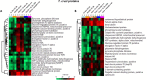
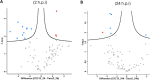
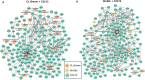
Trypanosoma cruzi is the protozoan that causes Chagas disease (CD), an endemic parasitosis in Latin America distributed around the globe. If CD is not treated in acute phase, the parasite remains silent for years in the host's tissues in a chronic form, which may progress to cardiac, digestive or neurological manifestations. Recently, studies indicated that the gastrointestinal tract represents an important reservoir for T. cruzi in the chronic phase. During interaction T. cruzi and host cells release extracellular vesicles (EVs) that modulates the immune system and infection, but the dynamics of secretion of host and parasite molecules through these EVs is not understood. Now, we used two cell lines: mouse myoblast cell line C2C12, and human intestinal epithelial cell line Caco-2to simulate the environments found by the parasite in the host. We isolated large EVs (LEVs) from the interaction of T. cruzi CL Brener and Dm28c/C2C12 and Caco-2 cells upon 2 and 24 h of infection. Our data showed that at two hours there is a strong cellular response mediated by EVs, both in the number, variety and enrichment/targeting of proteins found in LEVs for diverse functions. Qualitative and quantitative analysis showed that proteins exported in LEVs of C2C12 and Caco-2 have different patterns. We found a predominance of host proteins at early infection. The parasite-host cell interaction induces a switch in the functionality of proteins carried by LEVs and a heterogeneous response depending on the tissues analyzed. Protein-protein interaction analysis showed that cytoplasmic and mitochondrial homologues of the same parasite protein, tryparedoxin peroxidase, were differentially packaged in LEVs, also impacting the interacting molecule of this protein in the host. These data provide new evidence that the interaction with T. cruzi leads to a rapid tissue response through the release of LEVs, reflecting the enrichment of some proteins that could modulate the infection environment.
Keywords: Trypanosoma cruzi; Chagas disease; Extracellular vesicles; Mass spectrometry; Microvesicles.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures






References
-
- Chagas C. Nova tripanozomiaze humana: Estudos sobre a morfolojia e o ciclo evolutivo do Schizotrypanum cruzi n. gen., n. sp., ajente etiolojico de nova entidade morbida do homem. Mem. Inst. Oswaldo Cruz. 1909 doi: 10.1590/S0074-02761909000200008. - DOI
-
- PAHO (Pan-American Health Organization). Factsheet: Chagas Disease in the Americas for Public Health Workers. Accessed in May 2023.
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

