A cysteine-specific solubilizing tag strategy enables efficient chemical protein synthesis of difficult targets
- PMID: 38425513
- PMCID: PMC10901488
- DOI: 10.1039/d3sc06032b
A cysteine-specific solubilizing tag strategy enables efficient chemical protein synthesis of difficult targets
Abstract
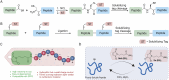
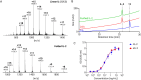
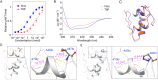
We developed a new cysteine-specific solubilizing tag strategy via a cysteine-conjugated succinimide. This solubilizing tag remains stable under common native chemical ligation conditions and can be efficiently removed with palladium-based catalysts. Utilizing this approach, we synthesized two proteins containing notably difficult peptide segments: interleukin-2 (IL-2) and insulin. This IL-2 chemical synthesis represents the simplest and most efficient approach to date, which is enabled by the cysteine-specific solubilizing tag to synthesize and ligate long peptide segments. Additionally, we synthesized a T8P insulin variant, previously identified in an infant with neonatal diabetes. We show that T8P insulin exhibits reduced bioactivity (a 30-fold decrease compared to standard insulin), potentially contributing to the onset of diabetes in these patients. In summary, our work provides an efficient tool to synthesize challenging proteins and opens new avenues for exploring research directions in understanding their biological functions.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures



Similar articles
-
Palladium-Assisted Removal of a Solubilizing Tag from a Cys Side Chain To Facilitate Peptide and Protein Synthesis.Org Lett. 2016 Jun 17;18(12):3026-9. doi: 10.1021/acs.orglett.6b01442. Epub 2016 Jun 8. Org Lett. 2016. PMID: 27268382
-
Solubilizing Trityl-Type Tag To Synthesize Asx/Glx-Containing Peptides.Chembiochem. 2019 Aug 16;20(16):2063-2069. doi: 10.1002/cbic.201900193. Epub 2019 Apr 18. Chembiochem. 2019. PMID: 31001897
-
Easy-to-Attach/Detach Solubilizing-Tag-Aided Chemical Synthesis of an Aggregative Capsid Protein.Angew Chem Int Ed Engl. 2018 Feb 19;57(8):2105-2109. doi: 10.1002/anie.201711546. Epub 2018 Jan 25. Angew Chem Int Ed Engl. 2018. PMID: 29316103
-
Development of Trityl Group Anchored Solubilizing Tags for Peptide and Protein Synthesis.Chembiochem. 2019 Aug 1;20(15):1906-1913. doi: 10.1002/cbic.201900105. Epub 2019 May 27. Chembiochem. 2019. PMID: 30810260 Review.
-
The Journey for the Total Chemical Synthesis of a 53 kDa Protein.Acc Chem Res. 2019 Dec 17;52(12):3361-3371. doi: 10.1021/acs.accounts.9b00372. Epub 2019 Sep 19. Acc Chem Res. 2019. PMID: 31536331 Review.
Cited by
-
Uncovering a Latent Bioactive Interleukin-6 Glycoform.Angew Chem Int Ed Engl. 2024 Dec 9;63(50):e202411213. doi: 10.1002/anie.202411213. Epub 2024 Oct 24. Angew Chem Int Ed Engl. 2024. PMID: 39103293 Free PMC article.
-
Peptide Multifunctionalization via Modular Construction of Trans-AB2C Porphyrin on Resin.Adv Sci (Weinh). 2025 Apr;12(14):e2409771. doi: 10.1002/advs.202409771. Epub 2025 Feb 19. Adv Sci (Weinh). 2025. PMID: 39973068 Free PMC article.
-
Selective Activation of Peptide-Thioester Precursors for Templated Native Chemical Ligations.Angew Chem Int Ed Engl. 2025 Jan 2;64(1):e202413644. doi: 10.1002/anie.202413644. Epub 2024 Oct 25. Angew Chem Int Ed Engl. 2025. PMID: 39198217 Free PMC article.
-
Advances in the chemical synthesis of human proteoforms.Sci China Life Sci. 2025 Apr 8. doi: 10.1007/s11427-024-2860-5. Online ahead of print. Sci China Life Sci. 2025. PMID: 40210795 Review.
References
LinkOut - more resources
Full Text Sources

