Prenatal diesel exhaust exposure alters hippocampal synaptic plasticity in offspring
- PMID: 38431308
- PMCID: PMC10968710
- DOI: 10.18632/aging.205592
Prenatal diesel exhaust exposure alters hippocampal synaptic plasticity in offspring
Abstract
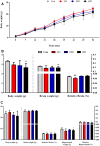
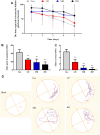
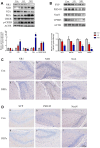
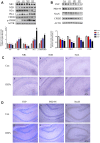
Diesel exhaust particles (DEPs) are major air pollutants emitted from automobile engines. Prenatal exposure to DEPs has been linked to neurodevelopmental and neurodegenerative diseases associated with aging. However, the specific mechanism by DEPs impair the hippocampal synaptic plasticity in the offspring remains unclear. Pregnant C57BL/6 mice were administered DEPs solution via the tail vein every other day for a total of 10 injections, then the male offsprings were studied to assess learning and memory by the Morris water maze. Additionally, protein expression in the hippocampus, including CPEB3, NMDAR (NR1, NR2A, NR2B), PKA, SYP, PSD95, and p-CREB was analyzed using Western blotting and immunohistochemistry. The alterations in the histomorphology of the hippocampus were observed in male offspring on postnatal day 7 following prenatal exposure to DEPs. Furthermore, 8-week-old male offspring exposed to DEPs during prenatal development exhibited impairments in the Morris water maze test, indicating deficits in learning and memory. Mechanistically, the findings from our study indicate that exposure to DEPs during pregnancy may alter the expression of CPEB3, SYP, PSD95, NMDAR (NR1, NR2A, and NR2B), PKA, and p-CREB in the hippocampus of both immature and mature male offspring. The results offer evidence for the role of the NMDAR/PKA/CREB and CPEB3 signaling pathway in mediating the learning and memory toxicity of DEPs in male offspring mice. The alterations in signaling pathways may contribute to the observed damage to synaptic structure and transmission function plasticity caused by DEPs. The findings hold potential for informing future safety assessments of DEPs.
Keywords: CPEB3; NMDA receptor; diesel exhaust particles; hippocampus; neurotoxicity.
Conflict of interest statement
Figures

Similar articles
-
Prenatal exposure to diesel exhaust particles causes anxiety, spatial memory disorders with alters expression of hippocampal pro-inflammatory cytokines and NMDA receptor subunits in adult male mice offspring.Ecotoxicol Environ Saf. 2019 Jul 30;176:34-41. doi: 10.1016/j.ecoenv.2019.03.090. Epub 2019 Mar 25. Ecotoxicol Environ Saf. 2019. PMID: 30921694
-
In utero exposure of mice to diesel exhaust particles affects spatial learning and memory with reduced N-methyl-D-aspartate receptor expression in the hippocampus of male offspring.Neurotoxicology. 2015 Sep;50:108-15. doi: 10.1016/j.neuro.2015.08.009. Epub 2015 Aug 18. Neurotoxicology. 2015. PMID: 26291742
-
Prenatal morphine alters the synaptic complex of postsynaptic density 95 with N-methyl-D-aspartate receptor subunit in hippocampal CA1 subregion of rat offspring leading to long-term cognitive deficits.Neuroscience. 2009 Feb 18;158(4):1326-37. doi: 10.1016/j.neuroscience.2008.11.007. Epub 2008 Nov 8. Neuroscience. 2009. PMID: 19041927
-
Developmental toxicity of diesel exhaust: a review of studies in experimental animals.Reprod Toxicol. 2013 Dec;42:1-17. doi: 10.1016/j.reprotox.2013.06.074. Epub 2013 Jul 4. Reprod Toxicol. 2013. PMID: 23831197 Review.
-
Hippocampal long-term synaptic plasticity and signal amplification of NMDA receptors.Crit Rev Neurobiol. 2006;18(1-2):71-84. doi: 10.1615/critrevneurobiol.v18.i1-2.80. Crit Rev Neurobiol. 2006. PMID: 17725510 Review.
References
-
- Fussell JC, Franklin M, Green DC, Gustafsson M, Harrison RM, Hicks W, Kelly FJ, Kishta F, Miller MR, Mudway IS, Oroumiyeh F, Selley L, Wang M, Zhu Y. A Review of Road Traffic-Derived Non-Exhaust Particles: Emissions, Physicochemical Characteristics, Health Risks, and Mitigation Measures. Environ Sci Technol. 2022; 56:6813–35. 10.1021/acs.est.2c01072 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

