Variety-dependent accumulation of glucomannan in the starchy endosperm and aleurone cell walls of rice grains and its possible genetic basis
- PMID: 38434111
- PMCID: PMC10905567
- DOI: 10.5511/plantbiotechnology.23.0809a
Variety-dependent accumulation of glucomannan in the starchy endosperm and aleurone cell walls of rice grains and its possible genetic basis
Abstract
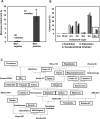
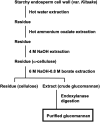
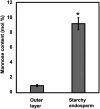
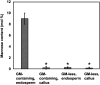
Plant cell wall plays important roles in the regulation of plant growth/development and affects the quality of plant-derived food and industrial materials. On the other hand, genetic variability of cell wall structure within a plant species has not been well understood. Here we show that the endosperm cell walls, including both starchy endosperm and aleurone layer, of rice grains with various genetic backgrounds are clearly classified into two groups depending on the presence/absence of β-1,4-linked glucomannan. All-or-none distribution of the glucomannan accumulation among rice varieties is very different from the varietal differences of arabinoxylan content in wheat and barley, which showed continuous distributions. Immunoelectron microscopic observation suggested that the glucomannan was synthesized in the early stage of endosperm development, but the synthesis was down-regulated during the secondary thickening process associated with the differentiation of aleurone layer. Significant amount of glucomannan in the cell walls of the glucomannan-positive varieties, i.e., 10% or more of the starchy endosperm cell walls, and its close association with the cellulose microfibril suggested possible effects on the physicochemical/biochemical properties of these cell walls. Comparative genomic analysis indicated the presence of striking differences between OsCslA12 genes of glucomannan-positive and negative rice varieties, Kitaake and Nipponbare, which seems to explain the all-or-none glucomannan cell wall trait in the rice varieties. Identification of the gene responsible for the glucomannan accumulation could lead the way to clarify the effect of the accumulation of glucomannan on the agronomic traits of rice by using genetic approaches.
Keywords: all-or-none trait; cell wall; cellulose synthase-like family; glucomannan; rice endosperm.
© 2023 Japanese Society for Plant Biotechnology.
Conflict of interest statement
Conflict of interestThe authors declare that they have no conflict of interest.
Figures



References
-
- Baba K, Sone Y, Kaku H, Misaki A, Shibuya N, Itoh T (1994) Localization of hemicelluloses in the cell walls of some woody plants using immuno-gold electron microscopy. Holzforschung 48: 297–300
-
- Bassett A, Hooper S, Cichy K (2021) Genetic variability of cooking time in dry beans (Phaseolus vulgaris L.) related to seed coat thickness and the cotyledon cell wall. Food Res Int 141: 109886. - PubMed
LinkOut - more resources
Full Text Sources
