Diallyl trisulfide inhibits osteosarcoma 143B cell migration, invasion and EMT by inducing autophagy
- PMID: 38434350
- PMCID: PMC10907726
- DOI: 10.1016/j.heliyon.2024.e26681
Diallyl trisulfide inhibits osteosarcoma 143B cell migration, invasion and EMT by inducing autophagy
Abstract
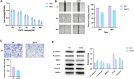
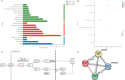
Background: Diallyl trisulfide (DATS), a compound derived from garlic, has been demonstrated its anti-cancer properties. While it has been shown to inhibit the expression of epidermal growth factor receptor (EGFR) in various cancers, its effects on osteosarcoma (OS) cells remain unclear. This study aimed to investigate the impacts of DATS on OS cells growth, migration, invasion, epithelial-mesenchymal transition (EMT) and autophagy, as well as its underlying mechanisms which was involving in the EGFR/PI3K/AKT/mTOR pathway.
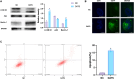
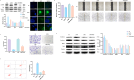
Methods: In this study, human osteosarcoma cells (143B) were treated with different concentrations of DATS (10, 50, 100 and 200 μM) for 24 and 48 h, respectively. Cell viability was measured using CCK8, the half lethal concentration was selected for the following experiments. Wound healing and transwell assays were performed to evaluate migration and invasion abilities, while flow cytometry was used to measure apoptosis. Quantitative reverse transcription polymerase chain reaction (qRT-PCR), Western blotting, and confocal imaging were employed to analyze the related mRNA and protein expression levels of epithelial-mesenchymal transition (EMT), EGFR/Phosphoinositide 3 kinase (PI3K)/AKT/Mammalian target of rapamycin (mTOR) signaling pathway and autophagy-related markers.
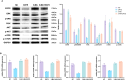
Results: DATS significantly inhibited proliferation, migration and EMT in osteosarcoma cells. Additionally, DATS promoted cell apoptosis and induced autophagy, which could be rescued by the autophagy inhibitor 3-methyladenine (3-MA). Moreover, DATS treatment led to the inactivation of the EGFR/PI3K/AKT/mTOR pathway in osteosarcoma cells.
Conclusions: This study demonstrated that DATS inhibited osteosarcoma cell growth, migration and EMT, but inducing apoptosis and autophagy. These effects were mediated by the inactivation of the EGFR/PI3K/AKT/mTOR signaling pathway. These findings suggested that DATS could serve as a potential therapeutic agent for osteosarcoma treatment.
Keywords: Autophagy; Diallyl trisulfide; EGFR/PI3K/AKT/mTOR; Osteosarcoma.
© 2024 The Authors.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures


Similar articles
-
The potential of diallyl trisulfide for cancer prevention and treatment, with mechanism insights.Front Cell Dev Biol. 2024 Sep 30;12:1450836. doi: 10.3389/fcell.2024.1450836. eCollection 2024. Front Cell Dev Biol. 2024. PMID: 39403128 Free PMC article. Review.
-
Diallyl trisulfide regulates cell apoptosis and invasion in human osteosarcoma U2OS cells through regulating PI3K/AKT/GSK3β signaling pathway.Histol Histopathol. 2020 Dec;35(12):1511-1520. doi: 10.14670/HH-18-299. Epub 2020 Dec 29. Histol Histopathol. 2020. PMID: 33372687
-
Diallyl thiosulfinate enhanced the anti-cancer activity of dexamethasone in the side population cells of multiple myeloma by promoting miR-127-3p and deactivating the PI3K/AKT signaling pathway.BMC Cancer. 2021 Feb 6;21(1):125. doi: 10.1186/s12885-021-07833-5. BMC Cancer. 2021. PMID: 33549034 Free PMC article.
-
RILP inhibits tumor progression in osteosarcoma via Grb10-mediated inhibition of the PI3K/AKT/mTOR pathway.Mol Med. 2023 Oct 3;29(1):133. doi: 10.1186/s10020-023-00722-6. Mol Med. 2023. PMID: 37789274 Free PMC article.
-
Insight Into the Role of Autophagy in Osteosarcoma and Its Therapeutic Implication.Front Oncol. 2019 Nov 15;9:1232. doi: 10.3389/fonc.2019.01232. eCollection 2019. Front Oncol. 2019. PMID: 31803616 Free PMC article. Review.
Cited by
-
Trends and future directions of autophagy in osteosarcoma: A bibliometric analysis.Open Med (Wars). 2024 Dec 3;19(1):20241080. doi: 10.1515/med-2024-1080. eCollection 2024. Open Med (Wars). 2024. PMID: 39655055 Free PMC article. Review.
-
Effects of Garlic on Breast Tumor Cells with a Triple Negative Phenotype: Peculiar Subtype-Dependent Down-Modulation of Akt Signaling.Cells. 2024 May 11;13(10):822. doi: 10.3390/cells13100822. Cells. 2024. PMID: 38786044 Free PMC article.
-
Aged Garlic Extract (AGE) and Its Constituent S-Allyl-Cysteine (SAC) Inhibit the Expression of Pro-Inflammatory Genes Induced in Bronchial Epithelial IB3-1 Cells by Exposure to the SARS-CoV-2 Spike Protein and the BNT162b2 Vaccine.Molecules. 2024 Dec 16;29(24):5938. doi: 10.3390/molecules29245938. Molecules. 2024. PMID: 39770027 Free PMC article.
-
The potential of diallyl trisulfide for cancer prevention and treatment, with mechanism insights.Front Cell Dev Biol. 2024 Sep 30;12:1450836. doi: 10.3389/fcell.2024.1450836. eCollection 2024. Front Cell Dev Biol. 2024. PMID: 39403128 Free PMC article. Review.
-
Interaction of ncRNAs and the PI3K/AKT/mTOR pathway: Implications for osteosarcoma.Open Life Sci. 2024 Aug 6;19(1):20220936. doi: 10.1515/biol-2022-0936. eCollection 2024. Open Life Sci. 2024. PMID: 39119480 Free PMC article. Review.
References
-
- Meyers P.A., Schwartz C.L., Krailo M.D., Healey J.H., Bernstein M.L., Betcher D., Ferguson W.S., Gebhardt M.C., Goorin A.M., Harris M., et al. Osteosarcoma: the addition of muramyl tripeptide to chemotherapy improves overall survival--a report from the Children's Oncology Group. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2008;26:633–638. doi: 10.1200/JCO.2008.14.0095. - DOI - PubMed
-
- Marina N.M., Smeland S., Bielack S.S., Bernstein M., Jovic G., Krailo M.D., Hook J.M., Arndt C., van den Berg H., Brennan B., et al. Comparison of MAPIE versus MAP in patients with a poor response to preoperative chemotherapy for newly diagnosed high-grade osteosarcoma (EURAMOS-1): an open-label, international, randomised controlled trial. Lancet Oncol. 2016;17:1396–1408. doi: 10.1016/S1470-2045(16)30214-5. - DOI - PMC - PubMed
-
- Gaspar N., Occean B.-V., Pacquement H., Bompas E., Bouvier C., Brisse H.J., Castex M.-P., Cheurfa N., Corradini N., Delaye J., et al. Results of methotrexate-etoposide-ifosfamide based regimen (M-EI) in osteosarcoma patients included in the French OS2006/sarcome-09 study. Eur. J. Cancer Oxf. Engl. 2018;88:57–66. doi: 10.1016/j.ejca.2017.09.036. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

