Characterization of cardiac fibroblast-extracellular matrix crosstalk across developmental ages provides insight into age-related changes in cardiac repair
- PMID: 38434619
- PMCID: PMC10904575
- DOI: 10.3389/fcell.2024.1279932
Characterization of cardiac fibroblast-extracellular matrix crosstalk across developmental ages provides insight into age-related changes in cardiac repair
Abstract
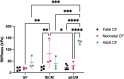
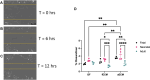
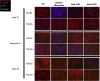
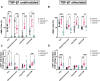
Heart failure afflicts an estimated 6.5 million people in the United States, driven largely by incidents of coronary heart disease (CHD). CHD leads to heart failure due to the inability of adult myocardial tissue to regenerate after myocardial infarction (MI). Instead, immune cells and resident cardiac fibroblasts (CFs), the cells responsible for the maintenance of the cardiac extracellular matrix (cECM), drive an inflammatory wound healing response, which leads to fibrotic scar tissue. However, fibrosis is reduced in fetal and early (<1-week-old) neonatal mammals, which exhibit a transient capability for regenerative tissue remodeling. Recent work by our laboratory and others suggests this is in part due to compositional differences in the cECM and functional differences in CFs with respect to developmental age. Specifically, fetal cECM and CFs appear to mitigate functional loss in MI models and engineered cardiac tissues, compared to adult CFs and cECM. We conducted 2D studies of CFs on solubilized fetal and adult cECM to investigate whether these age-specific functional differences are synergistic with respect to their impact on CF phenotype and, therefore, cardiac wound healing. We found that the CF migration rate and stiffness vary with respect to cell and cECM developmental age and that CF transition to a fibrotic phenotype can be partially attenuated in the fetal cECM. However, this effect was not observed when cells were treated with cytokine TGF-β1, suggesting that inflammatory signaling factors are the dominant driver of the fibroblast phenotype. This information may be valuable for targeted therapies aimed at modifying the CF wound healing response and is broadly applicable to age-related studies of cardiac remodeling.
Keywords: cardiac development; cardiac fibroblast; extracellular matrix; fibrosis; wound healing.
Copyright © 2024 Perreault, Daley, Watson, Rastogi, Jaiganesh, Porter, Duffy and Black.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Elastic, silk-cardiac extracellular matrix hydrogels exhibit time-dependent stiffening that modulates cardiac fibroblast response.J Biomed Mater Res A. 2016 Dec;104(12):3058-3072. doi: 10.1002/jbm.a.35850. Epub 2016 Aug 11. J Biomed Mater Res A. 2016. PMID: 27480328 Free PMC article.
-
Cardiac Fibroblast-Specific Activating Transcription Factor 3 Promotes Myocardial Repair after Myocardial Infarction.Chin Med J (Engl). 2018 Oct 5;131(19):2302-2309. doi: 10.4103/0366-6999.241794. Chin Med J (Engl). 2018. PMID: 30246716 Free PMC article.
-
Human iPSC-derived Committed Cardiac Progenitors Generate Cardiac Tissue Grafts in a Swine Ischemic Cardiomyopathy Model without Triggering Ventricular Arrhythmias.bioRxiv [Preprint]. 2024 Feb 17:2024.02.14.580375. doi: 10.1101/2024.02.14.580375. bioRxiv. 2024. PMID: 38405927 Free PMC article. Preprint.
-
Applications of Cardiac Extracellular Matrix in Tissue Engineering and Regenerative Medicine.Adv Exp Med Biol. 2018;1098:59-83. doi: 10.1007/978-3-319-97421-7_4. Adv Exp Med Biol. 2018. PMID: 30238366 Review.
-
Cardiac Fibroblasts and Myocardial Regeneration.Front Bioeng Biotechnol. 2021 Mar 25;9:599928. doi: 10.3389/fbioe.2021.599928. eCollection 2021. Front Bioeng Biotechnol. 2021. PMID: 33842440 Free PMC article. Review.
Cited by
-
Mechanism of action and experimental validation of key genes common to diabetic retinopathy and coronary heart disease based on multiple bioinformatics investigations.Front Genet. 2025 Mar 19;16:1548147. doi: 10.3389/fgene.2025.1548147. eCollection 2025. Front Genet. 2025. PMID: 40176795 Free PMC article.
-
The epigenetic regulation of crosstalk between cardiac fibroblasts and other cardiac cell types during stress.Front Cardiovasc Med. 2025 Apr 8;12:1539826. doi: 10.3389/fcvm.2025.1539826. eCollection 2025. Front Cardiovasc Med. 2025. PMID: 40264508 Free PMC article. Review.
References
LinkOut - more resources
Full Text Sources

